- 4192 Devonwood Way, Ashburn, Virginia, 20148
- Helpline: +1 (703) 665-3747
Recombinant Viral Vaccines Strategies for COVID-19
- Home
- Back to Journal
- Article Details
Introduction
During early December 2019, several patients were admitted to hospital in Wuhan, China with pneumonia-like symptoms. Deep sequencing identified the pathogenic agent as a novel coronavirus that was later termed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), [1]. This follows the emergence of two other novel coronavirus: SARS-CoV in 2003 (China) and Later Middle East respiratory syndrome (MERS: Saudi Arabia in 2012) in the 21st century, causing local endemics [2]. In January 2020, the virus started to spread rapidly throughout the population of Wuhan whilst presenting a fatality rate of 5.2% (Mortality Analyses- Johns Hopkins Coronavirus Resource Center, 2020). New cases were confirmed worldwide and by 30th January the World Health Organization (WHO) declared the outbreak a global emergency and the WHO elevated the status to a global Pandemic on 11th March 2020.
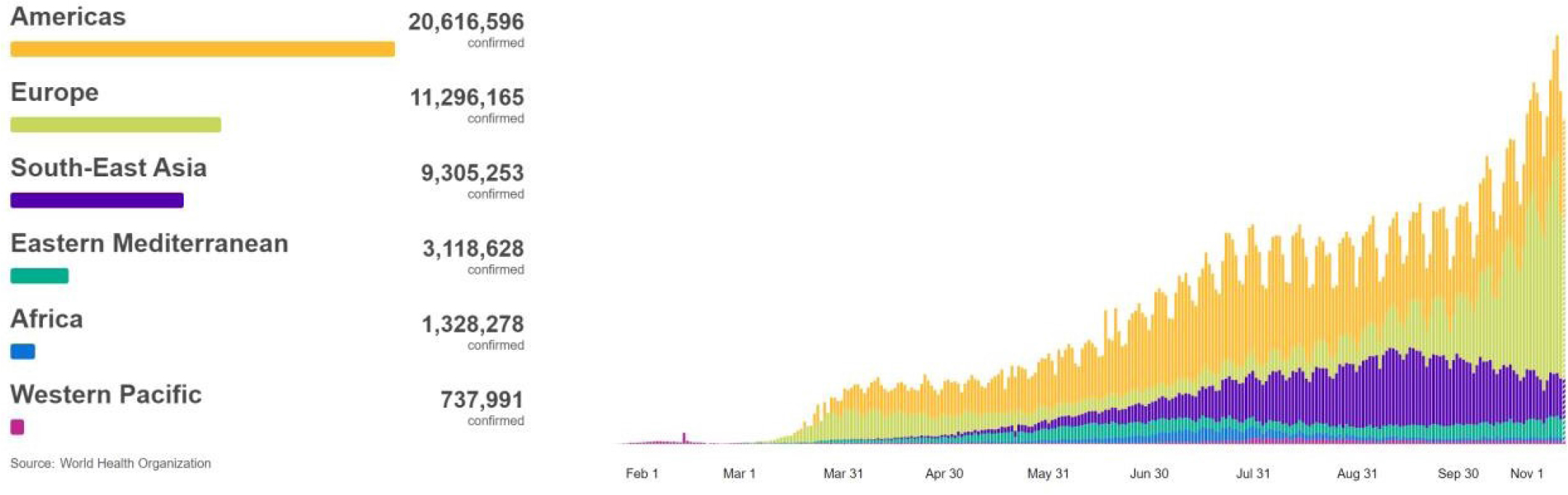
Figure 1: Graph illustrates the number of cases of coronavirus (COVID-19) by region globally.
Figure 1 is demonstrating the number of patients who have been diagnosed daily with COVID-19 from the 3rd January - 1st November 2020 Worldwide (WHO Coronavirus Disease (COVID-19) Dashboard, 2020).
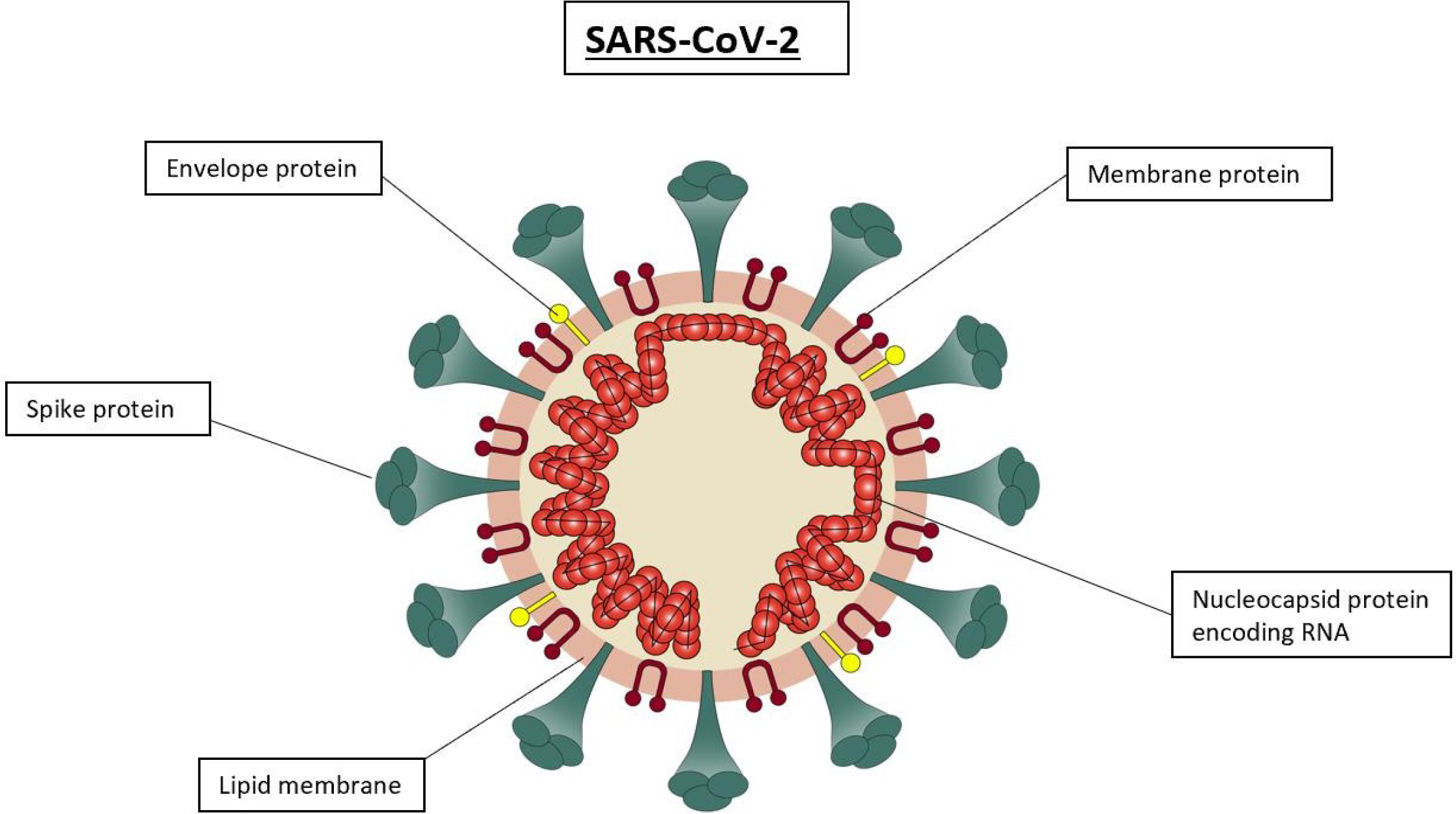
The SARS-CoV-2 virus can rapidly replicate in the respiratory tract, affecting primarily the lungs of the host. Although the onset of symptoms appears to be slow, the virus is highly transmittable during the asymptomatic phase [3]. As illustrated in figure 2, the virus consists of a positive sense single-stranded RNA genome; surround by an enveloped coat with the most prominent protein being the spike glycoprotein (S) which is a homotrimer composed of an S1 and S2 subunit. The S1 subunit forms the head of the spike and maintains a 3D structure in the Receptor Binding Domain (RBD) while the S2 subunit forms the stem anchoring the spike in the viral envelope [4]. The RBD of the S protein is a 394-glutamine residue which is identified by the lysine 31 residue on the angiotensin-converting enzyme 2 (ACE2) on the host cell for entry [5]. SARS-CoV- 2 enters host cell via the binding of ACE2 receptor on the cell membrane and the S protein, facilitating virus-cell membrane fusion and viral entry. Upon entry, the genomic RNA is released into the cytoplasm and translated into polymerase proteins, synthesized, and used to form copies of a genomic positive sense RNA [2].
Figure 2: Diagram representation of the SARS-CoV-2 structure causing the human coronavirus.
Figure 2 Portrays an enveloped, spherical virus which has a diameter of 120nm. The RNA genome is associated with the nucleoprotein which forms the nucleocapsid. Spike, envelope, and membrane proteins all form the outer layer of the virus.
ACE2 is a protein which is presented on the membranes of cells found in the lung, heart, arteries, intestine and kidneys [6]. It is essential in regulating intestinal functions and controlling the Renin-Angiotensin Aldosterone System (RAAS) pathway to regulate blood pressure and fluid balance as well as safeguarding organs from inflammatory injuries. The protease: TMPRSS2 slices ACE2 allowing SARS-CoV-2 entry into the host cell that alongside causing dysfunction leads to subsequent worsening of COVID-19 and finally initiate multi-organ failure [7].
Rapid vaccine development for SARS-CoV-2 was possible as the full genome sequence and structural information was made available to the public in record time allowing scientists around the world to begin research on different vaccines and therapeutic approaches immediately. Additionally, B-cell and T-cell epitope mapping, protein engineering, gene synthesis, and bioinformatics communications has helped predict suitable and potent targets against SARS-CoV-2 and provided the foundation to recombinant vaccine research. Furthermore, vaccine precision can be attained by knowing the atomic structure of the selected antigen and preserving the epitopes targeted by the vaccine [6].
In order to elicit an antibody-mediated immunity from a vaccine, choosing a correct antigen sequence which is conformational correct is essential [8]. Therefore, it is critical for the selected antigen to maintain the correct conformation of S protein which is a dynamic and metastable protein which holds two major conformational states, pre-fusion and post-fusion. The native pre-fusion spike protein is displayed on the surface of SARS-CoV-2 and is essential for viral entry. Upon connecting to ACE2 receptors on surface of host cell, protein rearrangement takes place as the virus and cell membrane fuse together and S protein conformational state changes to post-fusion [8,9]. The vaccine antigen is required to display the pre-fusion S protein contour and chemistry, in order to preserve the correct epitopes that are necessary in eliciting an increased neutralizing antibody response to the virus. An ideal vaccine will be formulated in a way that antigen delivery will influence T-cell functions and an effective response pattern where a balanced CD4+ and CD8+ T-cell response can be generated [10].
Typically, COVID-19 patients present with symptoms such as fever, cough, expectorating, loses of olfactory perception, shortness of breath and fatigue [11]. Following the death of the first COVID-19 patient, their pathology reports were examined by Xiao et al [7] which revealed lesions and fibrosis on the lungs whilst the lower respiratory tract and pulmonary alveolus were occupied with exudates. They hypothesized that SARS- CoV-2 primarily infects the lungs and intestines of the host through TMPRSS2-cleaved ACE2. Consequently, systemic inflammation and an abnormal immune response induce a cytokine storm resulting in loss of function in the cardiovascular system, respiratory system and other major organs [12].
All evidence and clinical data have led to the understanding that among COVID-19 patients, those with underlying diseases have a higher mortality rate [1,3,13]. Irregularity or loss of function of ACE2 further compromises the health of patients with diabetes, hypertension, and cardiovascular disease. Moreover, SARS-CoV-2 decreases the production of functional ACE2, which is why patients with underlying health diseases are more likely to develop severe and critical conditions [7].
Vaccine Strategy Design
Centers around the globe have conducted pre-clinical and clinical trials in hope of finding an effective COVID-19 preventative. Currently there are over 150 vaccine candidates in preclinical evaluation and 45 vaccine candidates in various stages of human clinical trials (Draft landscape of COVID-19 candidate vaccines, 2020) [14]. Many approaches have been conducted, such as live attenuated, inactivated virus vaccines, protein subunit vaccines, non-replicating viral vectors and nucleic acid vaccines. Live attenuated and inactivated vaccines are the most common, accounting for approximately one third of vaccines in the pipeline. However, these well-established methodologies fall predominantly outside of the scope of this review [15]. We reiterate these still form a key component of the arsenal of pipelines to address the current urgency of the pandemic.
There is a need to analyses and evaluate the vast amount of research currently ongoing for the multiple different recombinant vaccine platforms, in order to understand the weakness and strengths of each. In this current review, the structural characteristics, epidemiology, and pathogenicity of SARS-CoV-2 will be studied. Furthermore, the DNA, RNA and viral vectored vaccines in development for SARS-CoV, MERS, SARS-CoV-2, and other viral infections will be analyzed to identify the most efficacious and non-toxic vaccine platform for the prevention of SARS-CoV-2. In order to better understand what the best possible vaccine is for COVID-19, the current pipeline of recombinant vaccines will be appraised. Finally, we aim to consolidate the research of the literature regarding said approaches and identify any gaps in the ongoing pre-clinical and clinical trials.
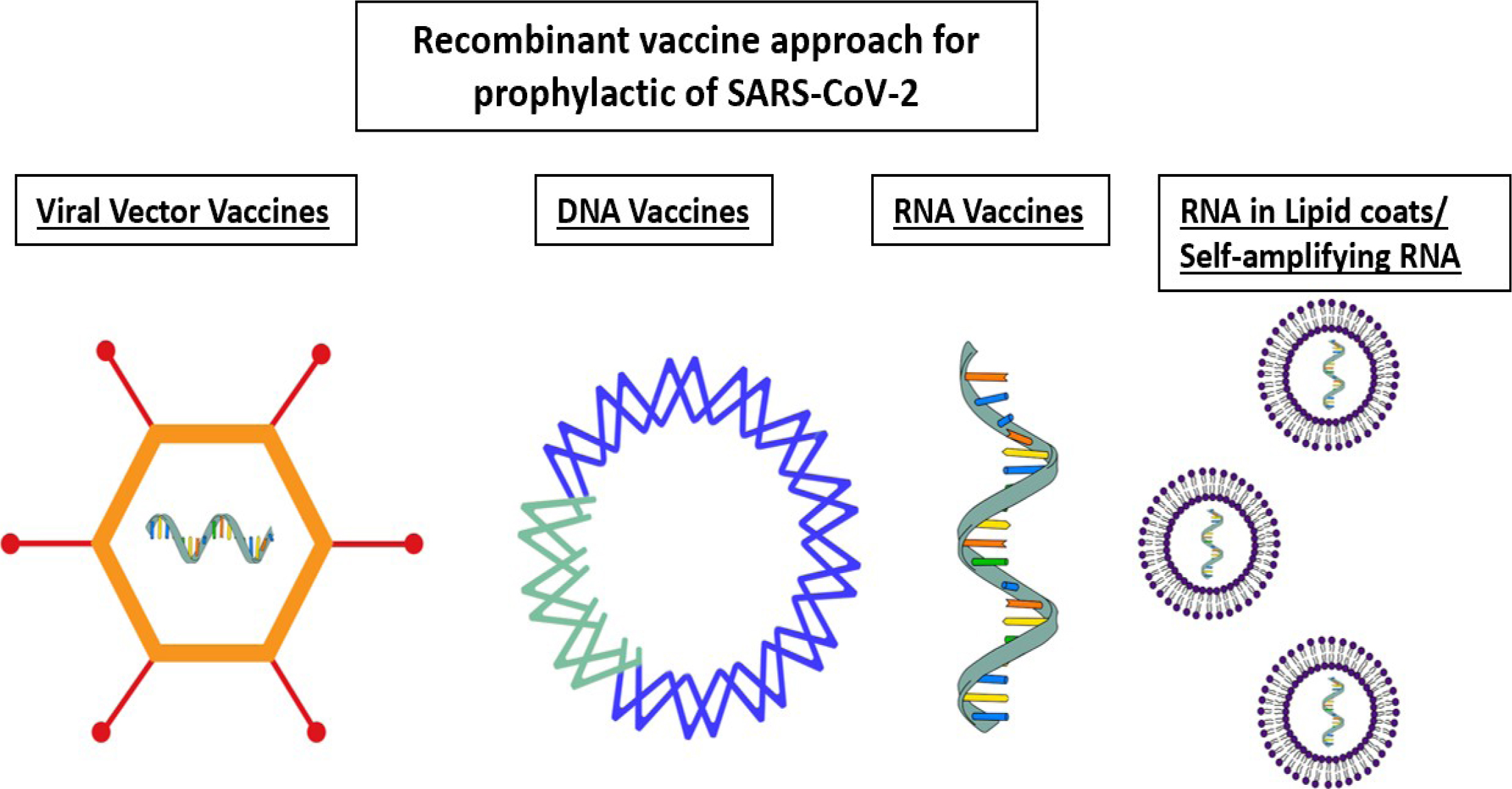
DNA vaccines can be formed by the spike gene of the SARS- CoV-2 encoded into a DNA plasmid such as that used by INOVIO pharmaceutical to form the INO-4800 vaccine. Similarly, the RNA encodes the spike protein and is injected into the human cells as Curevac are doing with their mRNA vaccine. The same RNA encoding S protein can be encapsulated in a lipid nanoparticle like Moderna’s vaccine. And finally, various types viral vector vaccines encoding the Spike protein such as ChAdOx1-S or the Adenovirus type5 vector.
Figure 3: Illustration of recombinant vaccine approaches for COVID-19.
Non-Replicating Viral Vectors
Non-replicating viral vectors act as a delivery vehicle where a non-pathogenic live virus carries a foreign protein into a vaccinated host which creates effective immunity against the selected antigen [16]. Oxford University partnering with AstraZeneca are currently developing the most advanced adenovirus vaccine candidate, a chimpanzee adenovirus vector ChAdOx1 (AZD1222) and it has reached stage 3 clinical trial (ISRCTN89951424).
Three other non-replicating viral vector candidates are also currently in final phases including CanSino Biological who are developing the Adenovirus type 5 vector NCT04540419, NCT04564716, NCT04505722.
The use of engineered mammalian viruses has been utilized for the development of non-replicating adenoviral vectors for a COVID-19 vaccine. The chimpanzee adenovirus vectored vaccine which was previously trialled for MERS-CoV expressing the S protein is now in clinical trials for the prophylactic of COVID-19. Another candidate is the adenovirus type 5 vector, being developed by CanSino biological who are currently in their Phase 3 clinical trials (NCT04540419). The Ad5 vectored COVID-19 vaccines encodes an optimized full-length spike gene. Furthermore, Johnson & Johnson have developed the natural virus called the Ad26-based vaccine to make a new adenoviral vector for COVID-19 and begun phase 3 clinical trials in September 2020 (NCT04505722). Ad26-based vaccine has previously been used for viruses such as HIV, RSV, Ebola and Zika virus. Nonetheless, potential drawbacks have been highlighted as the vaccine regimen for the previous viruses was a shot of Ad26-based vaccine plus a booster shot of a different vaccine which is not needed for COVID-19 therefore a direct comparison cannot be drawn. Additionally, some pre-existing immunity to Ad26 has been detected in parts of Africa and Southeast Asia (Johnson & Johnson Prepares to Resume Phase 3 ENSEMBLE Trial of its Janssen COVID-19 Vaccine Candidate in the U.S. [17].
Adenoviral vaccines are advantageous because of their fundamental adjuvant qualities, scalability, and broad tissue tropism. Gao et al [18], in their pre-clinical research have hypothesized that adenoviral based vaccine platforms can grant protection to those even with pre-existing adenoviral immunity however, results of the recent trials challenge this theory. Trials for Ad5 Vectored COVID-19 vaccine reported that participants with pre-existing Ad5 immunity demonstrated a slower immune response to SARS-CoV-2. Pre-existing Ad5 immunity lowered humoral immune response and affected the persistence of vaccine induced immunity negatively [19]. This proof in human clinical trials further validates the fact that results of pre-clinical trials are not always sustained in human models. Nonetheless, in previous studies heterologous or homologous prime boosters assisted the production of a stronger and more durable immunogenic response in those with pre-existing Ad5 immunity [20]. However, there is widespread concern regarding immunopathology and Antibody- Dependent Enhancement (ADE) events in relation to full-length spike glycoprotein vaccine antigens [21]. Therefore, the dose increase of Ad5 vectored vaccine could induce a higher chance of ADE amongst patients, but this concern can only be measured with further prolonged clinical studies.
Based on previous animal studies for coronavirus vaccines, the potential of ADE and vaccine associated respiratory disease have been an accumulating concern [22]. In order to reduce these risks occurring, high quality functional antibody responses and Th1-biased T-cell responses need to be induced by the vaccine. In a pre-clinical study for mRNA-1273 which was performed in parallel to the human clinical trials Corbett et al [23], reported that the structurally defined spike antigen induced a strong neutralizing activity. Furthermore, the gene- based delivery promoted Th1-biased response as well as a CD8+ T-cell response that protected the replication of the virus in lungs and nose of mice without traces of immunogenicity.
As shown in table 3, for ChAdOx1 vaccine, pre-existing adenovirus immunity was not an issue as the virus is based on a chimpanzee adenovirus with low seroprevalence in humans [24]. Notably, data suggests that a T-cell response is crucial in COVID-19 mitigation and those who acquired the virus in an asymptomatic manner developed a robust T-cell immunity [25]. ChAdOx1 vaccine demonstrated an increase in SARS- CoV-2 spike-specific effector T-cell response 7 days post vaccination. This rose by day 14 and was maintained for up to 56 days post-vaccination, which is in line with the current knowledge of adenovirus vectors. However, Folegatti et al [26] reported that the cellular response was not enhanced after the second dose of ChAdOx1. Future studies will need to demonstrate if an efficacious T-cell response and productivity from the vaccine can last longer than 56 days post-vaccination.
Nucleic Acid Vaccines
Historically, it was believed that DNA vaccines can provide higher stability over mRNA vaccines. However, the instability, half-life and immunogenicity of mRNA vaccines can be adapted and enhanced via established modification methods. For example, the recent development of a self-amplifying RNA (saRNA) vaccine technology by Imperial College of London to enhance the half-life of the RNA and improve the S protein expression level has now reached clinical trials (ISRCTN17072692) [27]. The vaccine has been trialled on 92 healthy volunteers and is now expanding to be given to over 200 people across London, UK. The saRNA approach directs the translation of antigen and viral replication which assists the intracellular RNA amplification and protein expression [28]. saRNA are typically derived from the genome backbone of an alphavirus, where the gene expressing the viral RNA replication mechanism remains intact. Those genes encoding viral structural proteins are substituted with the transgene expressing S protein antigen. As the saRNA is encapsulated in an LNP, it is able to elicit a substantially stronger immune response than an mRNA vaccine candidate [29].
In a study by Beissert et al [30], two strategies of RNA vectors are compared and analyzed. One strategy retains the replicase-encoding gene while the other encodes the antigen. Thus, the replicase structure is provided “in trans” by a saRNA or a non-replicating mRNA and facilitates the replication of the antigen encoding RNA. Results from the study demonstrated a robust and protective production of neutralizing antibody response in mice when immunized with antigen encoding RNA expressing hemagglutinin protein of influenza virus at nanogram doses. However, relatively high numbers of replicase-encoding RNAs were required. The study highlighted advantages of trans-amplifying approach when compared to conventional saRNA [30]. First highlighted benefit is safety, which stems from use of two separate RNAs. This averts the risk experienced with saRNA which are engineered to express budding-competent viral glycoproteins that could travel to extracellular vesicles and transfer to a new host cell. Secondly, the manufacturability benefit as an increase in production can be challenging for long RNA transcripts such as saRNAs. But in the trans-amplifying method the RNAs are shorter in length. However, there are two possible drawbacks to this approach such as the need to manufacture two RNA drugs and the additional complication in vivo delivery of both RNAs into the same cell in an efficient manner. Finally, trans- amplifying RNA approach could be improved by nucleoside modification, codon optimization, and stabilizing sequences, all of which are currently strategies implemented on mRNA vaccines but not yet possible for saRNA vaccines [30].
Plasmid DNA or synthetic RNA encoding the S protein Open Read Frame (ORF) is injected into the host. The RNA can be coated in a Lipid Nanoparticle (LNP) [31]. Research in small animal studies of DNA vaccine enabled the hypothesis that they could generate a broad immune response comparable with live-attenuated vaccines without the risk of pathogenicity [32]. However, human trials evaluating efficacy and safety of DNA vaccines conducted against influenza, hepatitis and malaria indicated poor immunogenicity [32]. There has been huge advances made by Moderna with mRNA vaccines as they have begun vaccine manufacturing process [33] (NCT04470427). Furthermore, INOVIO Pharmaceutical is developing a DNA vaccine (INO-4800) that is in stages 1/2 clinical trials (NCT04447781, NCT04336410).
The LNP encapsulated mRNA-1273 encoding the full-length S protein of SARS-CoV-2 is in development by Moderna [33]. Interest towards mRNA vaccines has been powered by the use of LNP, which stabilizes the mRNA, enhances the cellular uptake, and improves the bioavailability once inside the host cell [29]. Results of the mRNA-1273 trial demonstrate that a T-cell response was induced against the virus. Immune and non-immune cells were able to express the mRNA, leading to MHC class Class I I and MHC class II antigen presentation. Furthermore, the LNP enhanced delivery and efficacy by protecting the mRNA from degradation which enabled immunogenicity by the production of neutralizing antibody response and binding antibody responses against S-2P and RBD [33]. Moreover, the conformational stability of mRNA-1273 encoding the S-2P enables increased immunogenicity based on the preservation of neutralizing-sensitive epitopes at the peak of pre-fusion molecule (Jackson et al, 2020). As previously reported in Wrapp et al [8], presenting the native pre-fusion conformation of S protein to the immune system will elicit an efficient host antigen production and promote higher antibody response to SARS-CoV-2. Moderna have incorporated this into their mRNA-1273 vaccine.
A pre-clinical study by McKay et al (2020), included positive control (electroporated plasmid DNA) and a negative control (saRNA encoding rabies glycoprotein) to compare with the vaccine candidate (saRNA LNP). They also compared the neutralizing antibody titre results of their candidate vaccine in mice to serum obtained from recovered COVID-19 patients (Table 1), although levels of neutralizing antibody titres shown in mice are expected to be higher than those obtained in humans. Furthermore, as illustrated in table 1, the study by Espeseth et al [9] was the only one which challenged animals with an infection to see the protection level by the vaccine. Therefore, some knowledge of the protectively characteristic of the vaccine in animals would have been beneficial for researchers. SaRNA study as SARS-CoV-2 vaccine candidate by McKay et al [27] has progressed onto phase 1 of clinical trial studies for further investigation (ISRCTN17072692).
Currently, there are no licensed DNA or mRNA vaccines on the market and the safety profile in humans has not yet been fully approved. However, the potential benefits are adaptability, non-pathogenicity and development and manufacturing speed. These benefits have been confirmed in many pre-clinical and human clinical trials which have already been conducted for coronavirus and other infectious diseases [9,34,37]. Importantly, as well as producing an antibody and CD4+ T-cell response, they can also elicit an effective CD8+ cytotoxic T-cell response which could be crucial in the eradication of the infection [38]. This has been demonstrated in Smith et al [37] when INO- 4800 was used in pre-clinical trials, as a CD4+ but also CD8+ response was indicated at day 7 and continued to rise by day 10. Similarly, in the human clinical trials for Moderna’s mRNA-1273, considerable levels of CD4+ T-cell response was detected by day 29 and continued to rise by day 43. However, a lower CD8+ T-cell response was detected [33]. Conversely, in the study with Yang et al [36], while testing DNA vaccine candidates against SARS-CoV, they believed that the depletion of T-cells did not affect the rate of viral replication in their mouse animal model. Furthermore, they assumed that T-cells were unable to reduce viral replication in animals and hypothesized that T-cell immunity does not control the pulmonary virus replication in animal models. The results found in Yang et al [36] regarding the role of T-cells and viral replication contradicts the current understanding that T-cells like CD4+ are crucial in viral eradication [39].
Table 1: Comparison of pre-clinical trials of RNA and viral vectored vaccine on mice and non-human primates.
|
Effects of a SARS associated coronavirus vaccine in monkeys [18] Immunising rhesus macaques intramuscularly with a combination of three Ad5-SARS-CoV vectors or a Control vector and given a booster vaccination on day 28. |
Modified mRNA/lipid nanoparticle- based vaccines expressing respiratory syncytial virus F protein variants are immunogenic and protective in rodent models of RSV infection [9] |
Self-amplifying RNA SARS-CoV-2 lipid nanoparticle vaccine candidate induces high neutralizing antibody titres in mice [27] |
|
S1 specific Ab detected in monkeys |
High specific RSV F protein serum Ab in mice |
SARS-CoV-2 specific IgG levels observed in mice |
|
T-cell response observed in all animals |
High CD4 and CD8 T-cell response in all animals |
High cellular and cytokine response to SARS-CoV-2 |
|
Neutralizing Ab observed in all animals. Those with low pre-existing neutralising Ab to Ad5 showed similar response. |
High neutralising Ab titres observed in mice + non-human primates |
Higher neutralizing Ab observed in mice than patients who recovered from COVID- 19 |
|
Included negative control |
Included negative control |
Included positive and negative controls |
|
|
Vaccine protected all rats against RSV A+B challenge |
|
The left column in table 1 details the study for SARS-CoV, testing Ad5 vaccine on rhesus macaques and the middle is the study for RSV infection, testing an mRNA LNP vaccine candidate on rodent models. Fially, the right column is a study for SARS-CoV-2, analyzing effects of saRNA LNP vaccine in mice.
In other studies, by Muralidharan et al [40], animals that were immunized with whole-inactivated virus vaccines and later infected with RSV experienced allergic inflammation. When cytokine interleukin-4 production is heightened, mucus production, airway hyper responsiveness and other attenuated cytolytic T-cell activity collectively known as TH2 immune responses also increases. Therefore, it is assumed that minimizing the TH2- biased response and inducing a balanced host immune response to the pathogens is crucial in controlling and eliminating the infection [41].
Table 2: Comparison of two studies of DNA vaccines.
|
A DNA vaccine induces SARS coronavirus neutralization and protective immunity in mice [36] |
Immunogenicity of a DNA vaccine candidate for COVID-19 [37] |
|
Increase in T-cell CD4 Higher increase in T-cell CD8 |
Increase in T-cell CD4 Higher increase in T-cell CD8 |
|
Substantial Neutralising ab increase compared to control |
Substantial Neutralising ab increase compared to control |
|
Seroconversion – reduction of viral load in lungs and nasal turbines |
High level of S protein specific IgG Ab observed |
|
|
Production of blocking antibodies response to S protein |
The left column in table 2 explains details of a pre-clinical study of potential DNA vaccine for SARS-CoV experimenting on mice and non-human primates. The right column is a pre-clinical study testing INO-4800 (DNA plasmid vaccine) against SARS-CoV-2 on mice and non-human primates.
INOVIO pharmaceutical have developed a DNA plasmid vaccine which encodes the S protein and is delivered via electroporation. This technology was based on previous research with HIV, nipah virus, human papillomavirus and hepatitis B virus [37]. DNA vaccines induced immunopathology has been an issue of concern for SARS-CoV and MERS and now possibly SARS-CoV-2. Table 2 demonstrates that in pre-clinical trials Smith et al [37] have reported no evidence of immune pathogenesis in their mice or non-human primate models when immunized with INO-4800. Table 2 also highlights that in pre-clinical trials for DNA vaccines, a higher level of CD8+ was observed than CD4+ post immunization. Furthermore, it has been discussed previously in this review that high levels of CD8+ cytotoxic T-cell production could lead to vaccine induced immunopathology. Although the results in pre-clinical trials and animal models were promising, experience has proven that these results are not always sustained in human clinical trials. Subsequently, DNA and RNA vaccines are still in clinical trial stages and have not yet been licensed for use. However, DNA vaccines could hold some advantages over the traditional live attenuated or inactivated vaccines. Smith et al [37], report the stability characteristic of the INO-4800 vaccine and the ability for it to be stored at room temperature for up to 1 year. This feature is especially useful in context of the recent pandemic as it can be distributed and stockpiled across the world in all climates and environments.
Codagenix are developing an intranasal live-attenuated vaccine candidate called CDX-005. This candidate is engineered using codon optimization software platform which has enabled them to recode the genome of the SARS-CoV-2 virus by inserting mutations into it. The mutations results in the vaccine to be non-pathogenic however still stimulate a robust T-cell and antibody immune response that can mimic the wild-type virus.
Table 3: Comparison of 3 early phase human volunteer clinical trials for vaccine candidates of SARS-CoV-2.
|
Clinical Trial |
mRNA Vaccine against |
Recombinant adenovirus |
ChAdOx1 nCoV-19 vaccine against SARS-CoV-2 [26] |
|
Participants |
45 adult participants |
195 adult participants |
1077 adult participants |
|
Pre-existing immunity |
N/A |
Lower vaccine response observed in those with pre-existing immunity to adenovirus |
No pre-existing immunity detected |
|
Serology Tests |
No SARS_COV-2 serology screening for participants |
Serologically screened all participants for SARS-CoV-2 |
Serologically tested some participants for SARS-CoV-2 |
|
Dosage |
Trialled a low, medium and high dose |
Trialled a low, medium and high dose |
Trialled only 1 dose with and without prophylactic paracetamol |
|
Side Effects |
Demonstrated increased side effect after second vaccination with some reporting severe side effects with a higher dose |
Normal number of side effects. Not dose-dependant |
Higher side effects than control with some reporting sever. |
|
Control |
No control |
No control |
MenACWY as control |
Trials specified in table 3 are crucial elements of (left column) the phase 1, dose-escalation, open-label trial preliminary report of mRNA-1273. The dose-escalation, open-label, non-randomized, first-in- human trial of Ad-5 (Middle column). And finally the phase 1/2, single-blind, randomized controlled trial preliminary report of ChAdOx1 vaccine (Right column).
As displayed in Table 3, in a phase 1 human clinical trial of recombinant adenovirus type-5 vectored COVID-19 vaccine by Zhu et al [19], 108 participants were enrolled, and all were serologically tested. Only those who showed negative results in serum specific IgM and IgG antibodies for SARS-CoV-2 were included in the trial for the non-replicating Ad5 vectored COVID-19 vaccine. In the other phase 1 human clinical trial by Jackson et al [33], none of the participants were screened for SARS-CoV-2 infection by serology or PCR before enrolment as it was not part of the exclusion criteria. Finally, the last phase 1 human clinical trial in the current review was by Folegatti et al [26], volunteers with a history of laboratory confirmed SARS-CoV-2 infection were excluded from the study. Yet, there is no mention of serologically testing the large number of patients involved. Study protocol was later amended, and healthcare workers were recruited onto the study after presenting negative results in serological screening yet only 98 of 1077 participants were tested prior to enrolment.
Serology Testing
Evidence from human challenge studies and preclinical trials involving Rhesus Macaques indicate that infection of SARS-CoV-2 elicits a T-cell and antibody immune response which is protective for some period of time from re- infection [42]. In safety and efficacy trials for vaccine candidates, typically human participants will be those who are in a higher risk of infection, such as healthcare workers as this will produce results of higher statistical quality [43]. However, this might lead to enrolment of those who have been infected with COVID- 19 but in an asymptomatic manner and are therefore unaware. Serological testing of all clinical trial participants at the start will be crucial and enhance the reliability, value, and interpretability of the results. Therefore, it seems reasonable that participants who tested positive in serology be excluded from the trial as they may be protected from re-infection and thus the results may be inaccurate. Additionally, if a higher number of people who are already immune and have pre-existing antibody tires are in the treatment arm of the study, the artificial boost of the measured efficacy will be indicated.
Dosage
In the clinical trial for mRNA-1273, Jackson et al [33], report that although a strong binding antibody response and neutralizing antibody response was elicited in participants after the first vaccination, a higher pseudo virus neutralizing activity was observed after second vaccination. Therefore, they will be implementing a 100µg two- dose vaccination schedule given 28 days apart for further clinical trials. For Ad5 vectored COVID-19 vaccine Zhu et al [19], selected their lower dose (5?×?1010 viral particles) and middle dose (1?×?1011 viral particles) to be given in a single dose in future clinical trials. The selected doses demonstrated lower adverse events whilst eliciting a robust cellular and humoral response. The clinical trials for ChAdOx1 are exploring both a single-dose or two-dose administration regimen in their phase 2 and 3 trials [26]. This is because whilst a single-dose was able to induce a cellular and humoral response against SARS-CoV-2, the booster enhanced neutralizing antibody titres in participants. Although a two-dose regimen is supporting a higher neutralizing antibody response activity for mRNA-1273 and ChAdOx1 vaccine, it is difficult to guarantee that the population will adhere to this two-dose regimen and attend appointments days apart. Furthermore, it could provide an individual with false sense of security of being immune from the virus after only receiving the first vaccination. It could also increase chance of acquiring the disease in between doses which are approximately 28 days apart. In a study, the adherence of the UK population to a two-dose Hepatitis A and B was observed [44]. Results demonstrated that the rate of adherence and series completion for multi-dose vaccines were exceptionally low amongst adults in the UK. This behavioral characteristic presents a possible concern for companies suggesting a two-dose vaccination regimen, although the heightened public awareness over COVID-19 would likely mean higher uptakes.
Summary
To summarize the findings of the studies, we can conclude that nucleic acid vaccines could be the pioneering solution for this ongoing pandemic. The simplicity of their manufacturing process and merely the need to sequence a pathogen enables them to be a great vaccine candidate for such events [45]. However, because of the lack of experience and knowledge in how these vaccines perform in humans, the time period before the release of these candidates can be substantially longer than the traditional methods or viral vectors.
Amongst all recombinant vaccines evaluated in this review, ChAdOx1 developed by University of Oxford has been considered as the most suitable vaccine technology in the sense that it is the least toxic and most efficacious for the prevention of SARS-CoV-2 [26]. Researchers explored the idea of single-dose and two-dose vaccine for ChAdOx1 and upon completion of their phase 2 and start of their phase 3 clinical trial. It is a non-replicating virus which has shown to induce a robust immune response after a single dose without causing infection (ISRCTN89951424). Moreover, the Chimpanzee adenovirus has been extensively studied and its low prevalence in the human population means pre-existing immunity is not a concern so far [46]. Additionally, the safety profile has been confirmed previously on subjects from 1 week to 90 years of age, rendering it safe to be used in all age groups, even those with comorbidities (Yong et al, 2019) [47]. Furthermore, the phase 1&2 study by Folegatti et al [26] included 1077 participants. It has a larger group of participants than the other human clinical trials included in this review, which increases the reliability of the results.
The emergence of SARS-CoV-2 and its rapid spread has enforced the need for a quicker development process of vaccines and therapeutic candidates. Research and development on coronaviruses since SARS-CoV and MERS epidemics have accelerated the knowledge and understanding we have today of the pathogenesis, diagnosis, and epidemiology of SARS-CoV-2. All the current research on this new strain of coronavirus adds additional expertise to the scientific community and our understanding of it. Whilst it is crucial to accelerate the development of the trials, it is also essential to evaluate the overall risk-benefit aspect of this acceleration during unmet medical needs. Realistically, a COVID-19 vaccine could be a year away before it is widely available to the general public. Yet, the efforts made by researchers, volunteers and the general public have enabled a faster development process for a potential vaccine which otherwise could have taken over 10 years.
Acknowledgement
We would like to thank Dr Sarah Siverns and Jose Covas Lima C Campos for reading the manuscript.
References
- Huang C, Wang Y, Li X, Ren L, Zhao J, et al. (2020) Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. The Lancet 395: 497 506. [View]
- Shereen M, Khan S, Kazmi A, Bashir N, Siddique R (2020) COVID-19 infection: Origin, transmission, and characteristics of human coronaviruses. Journal of Advanced Research 24: 91 98. [View]
- Noor A, Maqbool F, Bhatti Z, Khan A (2020) Epidemiology of CoViD-19 Pandemic: Recovery and mortality ratio around the globe. Pakistan Journal of Medical Sciences 36(COVID19-S4): S79–S84. [View]
- Malla A, Shanmugaraj B, Ramalingam S (2020) Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2): An Emerging Zoonotic Respiratory Pathogen in Humans. Journal of Pure and Applied Microbiology 14: 931 936. [View]
- Sah R, Rodriguez-Morales A, Jha R, Chu D, et al. (2020) Complete Genome Sequence of a 2019 Novel Coronavirus (SARS-CoV-2) Strain Isolated in Nepal. Microbiology Resource Announcements 9: e00169 20. [View]
- Ahmed S, Quadeer A, McKay M (2020) Preliminary Identification of Potential Vaccine Targets for the COVID-19 Coronavirus (SARS-CoV-2) Based on SARS-CoV Immunological Studies. Viruses 12: 254. [View]
- Xiao L, Sakagami H, Miwa N (2020) ACE2: The key Molecule for Understanding the Pathophysiology of Severe and Critical Conditions of COVID-19: Demon or Angel? Viruses 12: 491. [View]
- Wrapp D, Wang N, Corbett K, Goldsmith J, Hsieh C, et al. (2020) Cryo- EM structure of the 2019-nCoV spike in the prefusion conformation. Science 367: 1260 1263. [View]
- Espeseth A, Cejas P, Citron M, Wang D, DiStefano D, et al. (2020) Modified mRNA/lipid nanoparticle-based vaccines expressing respiratory syncytial virus F protein variants are immunogenic and protective in rodent models of RSV infection. npj Vaccines 5: 16. [View]
- Tai W, He L, Zhang X, Pu J, Voronin D, et al. (2020) Characterization of the receptor- binding domain (RBD) of 2019 novel coronavirus: implication for development of RBD protein as a viral attachment inhibitor and vaccine. Cellular & Molecular Immunology 17: 613 620. [View]
- Huang X, Wei F, Hu L, Wen L, Chen K (2020) Epidemiology and Clinical Characteristics of COVID-19. Archives of Iranian Medicine 23: 268 271. [View]
- Mehta P, McAuley D, Brown M, Sanchez E, Tattersall R, et al. (2020) COVID-19: consider cytokine storm syndromes and immunosuppression. The Lancet 395: 1033 1034. [View]
- Wang F, Kream R, Stefano G (2020) An Evidence Based Perspective on mRNA-SARS-CoV-2 Vaccine Development. Medical Science Monitor 26: e924700 1–e924700 8. [View]
- https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines
- Mulligan M (2020) An Inactivated Virus Candidate Vaccine to Prevent COVID-19. JAMA 324: 943. [View]
- Lee N, Lee J, Park S, Song C, Choi I, Lee J (2012) A review of vaccine development and research for industry animals in Korea. Clinical and Experimental Vaccine Research 1: 18 34. [View]
- https://www.jnj.com/our-%20company/johnson-johnson-prepares-to-resume-phase-3-ensemble-trial-of-its-janssen-covid-19-vaccine-%20candidate-in-the-us
- Gao W, Tamin A, Soloff A, D’Aiuto L, Nwanegbo E, et al. (2003) Effects of a SARS-associated coronavirus vaccine in monkeys. The Lancet 362: 1895 1896. [View]
- Zhu F, Li Y, Guan X, Hou L, Wang W, et al. (2020) Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored COVID-19 vaccine: a dose-escalation, open-label, non-randomised, first-in-human trial. The Lancet 395: 1845 1854. [View]
- Dolzhikova I, Zubkova O, Tukhvatulin A, Dzharullaeva A, Tukhvatulina N, et al. (2017) Safety and immunogenicity of GamEvac-Combi, a heterologous VSV- and Ad5-vectored Ebola vaccine: An open phase I/II trial in healthy adults in Russia. Human Vaccines & Immunotherapeutics 13: 613 620. [View]
- Cao X (2020) COVID-19: immunopathology and its implications for therapy. Nature Reviews Immunology 20: 269 270. [View]
- Graham B (2020) Rapid COVID-19 vaccine development. Science 368: 945 946. [View]
- Corbett K, Edwards D, Leist S, Abiona O, Boyoglu-Barnum S, et al. (2020) SARS-CoV-2 mRNA Vaccine Development Enabled by Prototype Pathogen Preparedness. Nature 586: 567–571. [View]
- Jia W, Channappanavar R, Zhang C, Li M, Zhou H, et al. (2019) Single intranasal immunization with chimpanzee adenovirus- based vaccine induces sustained and protective immunity against MERS-CoV infection. Emerging Microbes & Infections 8: 760 772. [View]
- Weiskopf D, Schmitz K, Raadsen M, Grifoni A, Okba N, et al. (2020) Phenotype and kinetics of SARS-CoV-2-specific T cells in COVID-19 patients with acute respiratory distress syndrome. Science Immunology 5: eabd2071. [View]
- Folegatti P, Ewer K, Aley P, Angus B, Becker S, et al. (2020) Safety and immunogenicity of the ChAdOx1 nCoV-19 vaccine against SARS-CoV-2: a preliminary report of a phase 1/2, single-blind, randomised controlled trial. The Lancet 396: 467 478. [View]
- McKay P, Hu K, Blakney A, Samnuan K, Brown J, (2020) Self-amplifying RNA SARS-CoV-2 lipid nanoparticle vaccine candidate induces high neutralizing antibody titers in mice. Nature Communications 11: 3523. [View]
- Florindo H, Kleiner R, Vaskovich-Koubi D, Acúrcio R, Carreira B, et al. (2020) Immune-mediated approaches against COVID-19. Nature Nanotechnology 15: 630 645. [View]
- Fuller D, Berglund P (2020) Amplifying RNA Vaccine Development. New England Journal of Medicine 382: 2469 2471. [View]
- Beissert T, Perkovic M, Vogel A, Erbar S, Walzer K, et al. (2020) A Trans-amplifying RNA Vaccine Strategy for Induction of Potent Protective Immunity. Molecular Therapy 28: 119 128. [View]
- Zhang C, Maruggi G, Shan H, Li J (2019) Advances in mRNA Vaccines for Infectious Diseases. Frontiers in Immunology 10: 594. [View]
- Ferraro B, Morrow M, Hutnick N, Shin T, Lucke C, Weiner D (2011) Clinical Applications of DNA Vaccines: Current Progress. Clinical Infectious Diseases 53: 296 302. [View]
- Jackson L, Anderson E, Rouphael N, Roberts P, Makhene M, et al. (2020) An mRNA Vaccine against SARS-CoV-2 — Preliminary Report. New England Journal of Medicine 383: 1920 1931. [View]
- Chahal J, Fang T, Woodham A, Khan O, Ling J, et al. (2017) An RNA nanoparticle vaccine against Zika virus elicits antibody and CD8+ T cell responses in a mouse model. Scientific Reports 7: 252. [View]
- Pardi N, Hogan M, Porter F, Weissman D (2018) mRNA vaccines — a new era in vaccinology. Nature Reviews Drug Discovery 17: 261 279. [View]
- Yang Z, Kong W, Huang Y, Roberts A, Murphy B, et al. (2004) A DNA vaccine induces SARS coronavirus neutralization and protective immunity in mice. Nature 428: 561 564. [View]
- Smith T, Patel A, Ramos S, Elwood D, Zhu X, et al. (2020) Immunogenicity of a DNA vaccine candidate for COVID-19. Nature Communications 11: 2601. [View]
- Shin, M., Shukla, S., Chung, Y., Beiss, V., Chan, S., Ortega-Rivera, O., Wirth, D., Chen, A., Sack, M., Pokorski, J. and Steinmetz, N., 2020. COVID-19 vaccine development and a potential nanomaterial path forward. Nature Nanotechnology,.
- Maloy K, Burkhart C, Junt T, Odermatt B, Oxenius A, et al. (2000) Cd4+ T Cell Subsets during Virus Infection. Journal of Experimental Medicine 191: 2159 2170. [View]
- Muralidharan A, Li C, Wang L, Li X (2016) Immunopathogenesis associated with formaldehyde-inactivated RSV vaccine in preclinical and clinical studies. Expert Review of Vaccines 16: 351 360. [View]
- Graham B, Henderson G, Tang Y, Lu X, Neuzil K, (1993) Priming immunization determines T helper cytokine mRNA expression patterns in lungs of mice challenged with respiratory syncytial virus. The Journal of Immunology 151: 2032 2040. [View]
- Bao L, Deng W, Gao H, Xiao C, Liu J, et al. (2020) Lack of Reinfection in Rhesus Macaques Infected with SARS-CoV-2. bioRxiv Preprint. [View]
- Lipsitch M, Kahn R, Mina M (2020) Antibody testing will enhance the power and accuracy of COVID-19- prevention trials. Nature Medicine 26: 818 819. [View]
- Johnson K, Lu X, Zhang D (2019) Adherence to hepatitis A and hepatitis B multi-dose vaccination schedules among adults in the United Kingdom: a retrospective cohort study. BMC Public Health, 19: 404. [View]
- Shi Y, Wang N, Zou Q (2020) Progress and challenge of vaccine development against 2019 novel coronavirus (2019-nCoV). Chinese journal of preventive medicine 54: 614 619. [View]
- Antrobus R, Coughlan L, Berthoud T, Dicks M (2014) Clinical Assessment of a Novel Recombinant Simian Adenovirus ChAdOx1 as a Vectored Vaccine Expressing Conserved Influenza A Antigens. Molecular Therapy 22: 668 674. [View]
Article Type
Review Article
Publication history
Received: November 02, 2021
Accepted: November 23, 2021
Published: November 30, 2021
Citation:
Parsi P, Collier W, Gould P (2021) Recombinant Viral Vaccines Strategies for COVID-19. Clar J Infect Dis Ther 02(01): 99–110.
Pardis Parsi1, William Collier2 and Phillip Gould1,2,3*
1School of Life Sciences, Coventry University, CV1 2DS, UK
2OVO Biomanufacturing Ltd. CV8 2EN, UK
3CSELS Coventry University, CV1 2DS, UK
*Corresponding author
Phillip Gould,
School of Life Sciences,
Coventry University,
CV1 2DS, and OVO Biomanufacturing Ltd.
CV8 2EN, and CSELS Coventry University,
CV1 2DS. UK;