- 4192 Devonwood Way, Ashburn, Virginia, 20148
- Helpline: +1 (703) 665-3747
Severe Covid-19 Disease – An Unusual Type of Skin Mottling
- Home
- Back to Journal
- Article Details
Abstract
Importance: Skin mottling in septic shock usually affects legs and arms and reflects significantly reduced skin perfusion. In the majority of most severely ill COVID-19 patients, we observed an unusual type of skin mottling mainly affecting the soles of feet.
Objective: To describe an unusual type of mottling occurring at the soles of feet in COVID-19 patients.
Design, setting, and participants: Retrospective, observational study of 18 COVID-19 patients treated in a 12-bed high dependency intensive care unit between March and May 2020. Patients were divided according to severity of respiratory failure into an invasive mechanical ventilation group (IV-G) and a non-invasive ventilation group (NIV-G).
Exposures: All COVID-19 Patients treated in the intensive care unit until discharge or dead.
Main outcomes and measures: Incidence, timely appearance and duration of skin mottling.
Results: There were no differences in demographic-, comorbidity data and SAPS III score at admission between groups. IV-G patients developed significantly higher SOFA-Scores, needed prolonged ventilation and had a significantly longer ICU stay. 80% of patients in the IV-G demonstrated symmetrical mottling at the soles of feet around time of intubation while only one patient in the NIV-G (14%) was affected. Patients in the IV-G demonstrated a highly augmented inflammatory response and coagulation activation resulting in significantly elevated maximum CRP, PCT, IL-6 and D-Dimer plasma concentrations in comparison with NIV-G. After initiation of therapeutic anticoagulation, skin mottling disappeared within one to two days.
Conclusion and relevance: Severely ill COVID-19 patients may develop a type of skin mottling usually not seen in other types of sepsis. At present, one can only speculate why mottling did not affect other skin areas. However, the observation of rapid improvement and disappearance of skin mottling after starting therapeutic anticoagulation in IV-G patients suggests a pathophysiologic link between local coagulation activation and the appearance of skin mottling at the soles of the feet.
Key Points
Question: Is there a rare, unusual type of skin mottling in patients suffering from severe COVID-19?
Findings: In this retrospective observational study, the majority of severely ill COVID-19 patients (80%) exhibited a rare type of skin mottling involving the soles of feet around the time of intubation. After starting therapeutic anticoagulation mottling disappeared within 24–48 hours.
Meaning: In severely ill COVID-19 patients skin mottling at the soles of feet disappeared after starting therapeutic anticoagulation suggesting a pathophysiologic link between local coagulation activation and the appearance of mottling at the soles of the feet.
Introduction
On January 7, 2020, the World Health Organization (WHO) officially announced a novel coronavirus to be responsible for an outbreak of severe pneumonia in the province of Wuhan, China [1]. Since then the virus rapidly spread and on March 11, 2020, the WHO declared COVID-19 a pandemic [2].
During the winter season the county of Landeck (Tirol), Austria, is a very popular skiing region crowded with tourist from all over the world. Iceland, Norway, Germany and Denmark were the first countries to report an increased number of SARS-CoV-2 infected people who had previously visited Ischgl, a famous village in the area [3]. On March 8, an employee of a popular après-ski bar tested positive for COVID-19. Detailed contact tracing suggested a superspreading event originating in Ischgl as primary cause of the rapid spread of COVID-19 in the county of Landeck.
In this area, the incidence of SARS-CoV-2 infected persons was extremely high approaching 1800 cases/100.000 inhabitants, respectively. The St. Vinzenz hospital is located centrally in in this county. During the pandemic, 139 patients were admitted because of severe COVID-19 symptoms. Twenty-five patients were transferred to our 12 bed interdisciplinary intensive care unit.
In the majority of patients with progressive ventilator failure treated with intubation and mechanical ventilation, we observed a very unusual type of skin mottling mainly located at the soles of the feet. In this retrospective, observational study, we report on type and incidence of transient skin lesions observed around the time of intubation and shortly thereafter.
Material and Methods
The interdisciplinary intensive care unit of the St. Vinzenz hospital consists of 12 high dependency ICU-beds, which were solely dedicated to the treatment of COVID-19 patients at the culmination of the pandemic (from March 10 to April 30, 2020). Overall, 25 of 139 hospitalized COVID-19 patients were admitted to ICU treatment. Seven patients were transferred to another hospital after initial stabilization and intubation. Two patients because of progressive respiratory failure necessitating ECMO therapy and five patients because of lacking ICU capacity. Of the remaining 18 patients, eleven patients needed intubation and mechanical ventilation. Seven patients responded to non-invasive ventilation or CPAP-therapy.
For analysis, we divided patients according to severity of respiratory failure into two groups: Intubated patients with invasive mechanical ventilation (IV-G; n=11) and patients responding to non-invasive ventilation (NIV-G; n=7). We extracted laboratory and clinical data from ICU-electronic records. Photo documentation of skin lesions were performed during daily clinical examination. Because of the retrospective, observational study design, informed patient consent was waived.
Comparisons between groups were made using Mann-Whitney-U-tests and Chi2-tests, as appropriate. Data in tables are presented as median, interquartile range, minimum and maximum values.
Results
There were no differences in demographic-, comorbidity data and SAPS III score at admission between groups (Table 1). However, patients with invasive mechanical ventilation developed significantly higher SOFA-Scores, needed prolonged ventilation and had a significantly longer ICU stay. 44% in the IV-G developed acute kidney injury necessitating veno-venous hemofiltration. ICU-mortality in IV-G patients was 18%; in the NIV-G mortality was 14% resulting in an overall ICU mortality of 16.6% (Table 1). All ICU-survivors left the hospital alive.
Table 1: Comparison of demography, comorbidity and ICU-data of COVID-19 patients with invasive and non-invasive ventilation.
|
|
|
INVASIVE VENTILATION |
|
NON-INVASIVE VENTILATION |
|||||||
|
|
n (%) |
Median |
IQR |
Min |
Max |
n (%) |
Median |
IQR |
Min |
Max |
p-value |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
DEMOGRAPHY |
|
|
|
|
|
|
|
|
|
|
|
|
age |
|
71 |
64–72 |
53 |
84 |
|
61 |
48–76 |
29 |
84 |
0.33 |
|
male sex |
10 (91) |
|
|
|
|
4 (57) |
|
|
|
|
0.09 |
|
BMI |
|
28 |
26–29 |
24.5 |
35.5 |
|
25.9 |
22–28 |
20 |
43 |
0.085 |
|
Incubation period |
10 |
7 |
4.5–7 |
1 |
33 |
7 |
6 |
5–7 |
5 |
7 |
0.32 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
COMORBIDITY |
|
|
|
|
|
|
|
|
|
|
|
|
hypertension |
7 (64) |
|
|
|
|
2 (29) |
|
|
|
|
0.15 |
|
CHD |
6 (55) |
|
|
|
|
2 (29) |
|
|
|
|
0.28 |
|
Chronic AF |
3 (27) |
|
|
|
|
1 (14) |
|
|
|
|
0.52 |
|
COPD |
1 (9) |
|
|
|
|
3 (43) |
|
|
|
|
0.09 |
|
Chronic renal failure |
1 (9) |
|
|
|
|
1 (14) |
|
|
|
|
0.73 |
|
Diabetes mellitus |
1 (9) |
|
|
|
|
0 |
|
|
|
|
0.41 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
ICU-DATA |
|
|
|
|
|
|
|
|
|
|
|
|
SAPS III |
|
62 |
56–65 |
49 |
73 |
|
51 |
47–71 |
44 |
81 |
0.21 |
|
SOFA max |
|
13 |
11–15 |
10 |
18 |
|
4 |
3–8 |
3 |
11 |
< 0.001 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
ICU-stay (days) |
|
33 |
25–43 |
19 |
53 |
|
5 |
4–7 |
2 |
8 |
< 0.001 |
|
MV (days) |
|
18 |
17–30 |
15 |
39 |
|
0 |
|
|
|
|
|
NIV (days) |
|
1 |
0–3 |
0 |
22 |
|
2 |
0–6 |
0 |
7 |
0.659 |
|
CPAP (days) |
|
4 |
0–6 |
0 |
10 |
|
0 |
0–4 |
0 |
5 |
0.246 |
|
Hemofiltration |
6 (54) |
22 |
10.5–31 |
6 |
37 |
|
0 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
MOTTLING |
|
|
|
|
|
|
|
|
|
|
|
|
Soles of feet |
9 (82) |
|
|
|
|
1 (14) |
|
|
|
|
0.005 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
OUTCOME |
|
|
|
|
|
|
|
|
|
|
|
|
survivor |
9 (82) |
|
|
|
|
|
6 (86) |
|
|
|
|
|
non-survivor |
2 (18) |
|
|
|
|
|
1 (14) |
|
|
|
|
BMI: Body Mass Index; CHD: Coronary Heart Disease; COPD: Chronic Obstructive Pulmonary Disease; SAPS: Simplified Acute Physiologic Score; SOFA max: Maximal Sequential Organ Failure Score; MV: Invasive Mechanical Ventilation; NIV: Non-Invasive Mechanical Ventilation; CPAP: Continuous Positive Pressure Ventilation
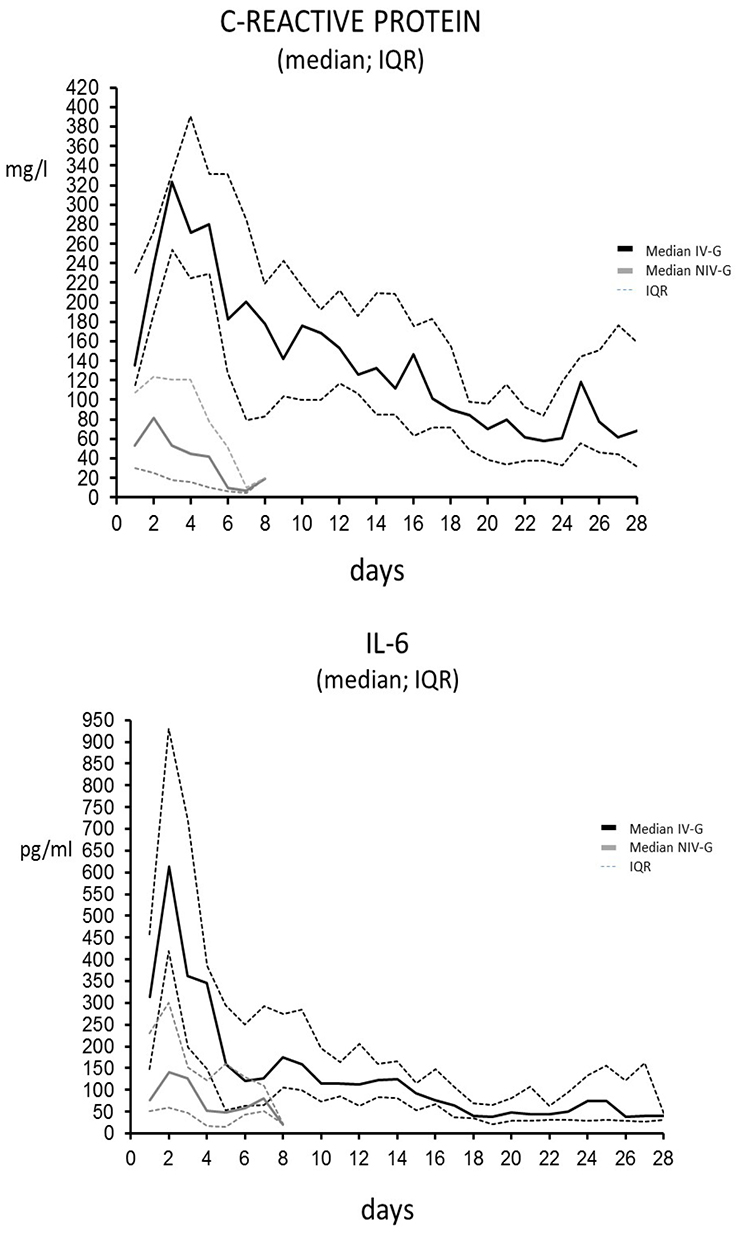
Table 2 shows laboratory data indicating inflammatory and coagulation activation in COVID-19 patients. Patients in the IV-G demonstrated a largely augmented inflammatory response and coagulation activation resulting in significantly elevated maximum CRP, PCT, IL-6 and D-Dimer plasma concentrations. There were no differences in median thrombocyte count although single patients in the IV-G showed moderate thrombocytopenia at admission.
Table 2: Laboratory data indication inflammatory and coagulation activation in COVID-19 patients with invasive and non-invasive ventilation.
|
|
Reference values |
INVASIVE VENTILATION |
NON-INVASIVE VENTILATION |
|||||||
|
|
|
Median |
IQR |
Min |
Max |
Median |
IQR |
Min |
Max |
p-value |
|
Leucocytes |
3.98–10.04 |
22.04 |
12.55–23.33 |
8.11 |
27.74 |
8.33 |
6.52–9.13 |
6.42 |
14.92 |
0.002 |
|
Lymphocytes |
1320–3570 |
400 |
300–560 |
200 |
1160 |
920 |
400–1000 |
380 |
1110 |
0.085 |
|
Ferritin |
30–400 µg% |
2955 |
2054–3245 |
688 |
3728 |
1648 |
562–3381 |
474 |
5480 |
0.301 |
|
CRP |
< 5mg/l |
412.4 |
323–501 |
241 |
529 |
114 |
20–154 |
12 |
190 |
< 0.001 |
|
PCT |
< 0.5 ng/ml |
2.66 |
1.25–10.3 |
0.69 |
13.9 |
0.28 |
0.22–0.54 |
0.12 |
2.24 |
< 0.001 |
|
Interleukin-6 |
< 7pg/ml |
1075 |
626–1795 |
270 |
2197 |
157 |
63–457 |
27,6 |
1915 |
0.008 |
|
|
|
|
|
|
|
|
|
|
|
|
|
D-Dimer |
< 0.5 mg/l |
9.35 |
3.9–16.7 |
1.97 |
80 |
2.44 |
0.95–5.21 |
0.55 |
5.55 |
0.006 |
|
Thrombocytes |
182–369 |
141 |
127–196 |
53 |
330 |
150 |
110–263 |
109 |
285 |
0.86 |
CRP: C-reactive protein; PCT: Procalcitonin
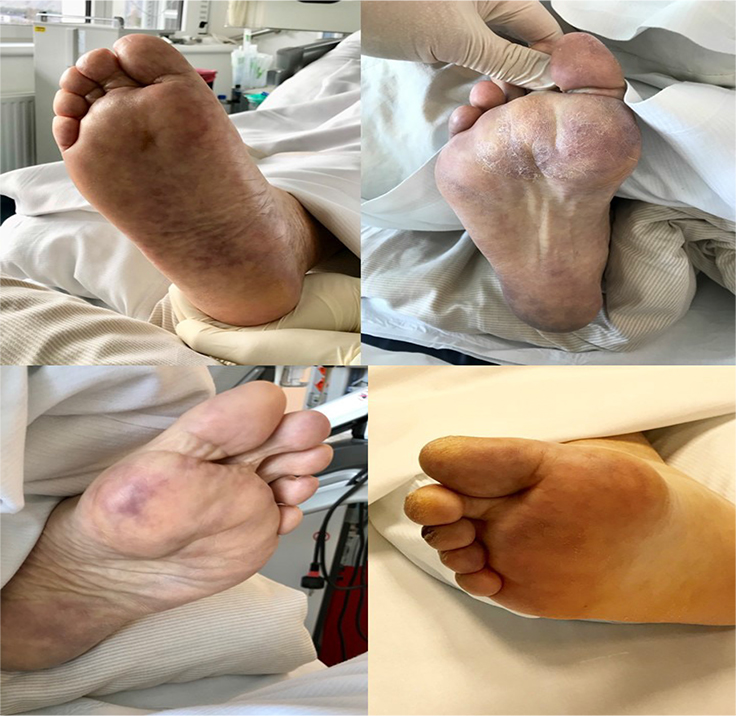
Figure 1 illustrates examples of skin mottling at the soles of the feet. The majority (n=9; 80%) of patients in the IV-G demonstrated this unusual type of mottling around time of intubation. Skin mottling always appeared symmetrically. Mottling persisted in most patients approximately 24h-48h hours. One patient in the NIV-G, who subsequently died, showed skin mottling at admission.
Figure 1: Examples of skin mottling at the soles of feet in patients with invasive mechanical ventilation.
Discussion
We report an unusual skin area affected by mottling in patients suffering from very severe COVID-19. In these patients skin mottling at the soles of the feet was present in nine of 11 IV-G patients (80%) around the time of intubation lasting 24–48 hours thereafter.
In septic shock, skin mottling usually affects legs and arms and often shows up most pronounced over the area of the knee [4]. Measurements of regional blood flow with laser-Doppler and skin observations using hyperspectral camera systems demonstrate a severely reduced skin blood flow and low blood oxygen saturation in affected areas [5]. Although the pathophysiology of skin mottling is not entirely clear, endothelial dysfunction, excessive adrenergic activity and activation of the coagulation system have all been discussed [6]. In clinical practice skin mottling involving legs and arms usually appears after prolonged shock periods often in combination with severe cardiac dysfunction.
On transthoracic echocardiography only mild left ventricular dysfunction was observed in 5 patients (45%) in the IV-G and 1 (14%) patient in the NIV-G (Chi2 Test p=0.17). No patient showed echocardiographic signs of overt hypovolemia. Therefore, prolonged cardiovascular failure is an unlikely explanation for this type of mottling.
We already know that vascular endothelial cells in different tissues are strongly affected by the SARS-CoV-2 [6]. The virus enters the endothelium via angiotensin-converting-enzyme II receptor (ACE-II-R), a receptor expressed on this type of cells [7]. The consumption of ACE-II-R during cell infection may lead to local vasoconstriction by increased presence of angiotensin II. Furthermore, endothelial cell activation during infection may promote a procoagulatory vascular state [7]. Autopsy studies have demonstrated diffuse endothelial damage in conjunction with widespread microvascular thrombosis in lungs of COVID-19 non-survivors [8]. In addition, a high incidence of pulmonary emboli in patients with COVID-19 has been reported [9]. Because of the highly elevated D-Dimer plasma concentrations in IV-G patients, we early started therapeutic anticoagulation (Table 2). Low molecular-weight heparins were given twice daily to achieve an Anti-Xa activity between 0.6 and 1 Anti-Xa IU/ml. After initiation of therapeutic anticoagulation, skin mottling disappeared in most patients within one to two days. Therefore, we speculate that increased formation of micro thrombi in the soles of feet might have played a key role in the pathogenesis of this unusual type of mottling.
It is interesting to note that overall mortality of COVID-19 patients was low, despite that fact that the population demonstrated a highly exaggerated inflammatory response demonstrated by IL-6 plasma concentrations, which by far exceed values, reported in different COVID-19 studies [10]. Small observational studies already suggest that patients with severe COVID-19 might benefit from early anticoagulation and probably local or systemic fibrinolytic therapy [11, 12, 13].
In summary, severely ill COVID-19 patients may develop a type of skin mottling usually not seen in septic shock. At present, one can only speculate why mottling did not affect other skin areas. However, the observation of rapid improvement and disappearance of skin mottling after starting therapeutic anticoagulation in IV-G patients suggests a pathophysiologic link between local coagulation activation and the appearance of skin mottling at the soles of the feet.
References
- Zhu N, Zhang D, Wang W, et al. (2020) China Novel Coronavirus Investigating and Research Team. A novel coronavirus from patients with pneumonia in China. N Engl J Med 382: 727–733. [View]
- https: //www.euro.who.int/en/health-topics/health-emergencies/coronavirus-covid-19/novel-coronavirus-2019-ncov
- Correa-Martinez CL, Kampmeier S, Kümpers P, et al. (2020) A pandemic in times of global tourism: superspreading and exportation of COVID-19 cases from a ski area in Austria. J Clin Microbiol 58: e00588–20. [View]
- Hariri G, Joffre J, Leblanc G, et al. (2019) Narrative review: clinical assessment of peripheral tissue perfusion in septic shock. Ann Intensive Care 13: 37. [View]
- Kazune S, Caica A, Suba O, et al. (2019) Relationship of mottling score, skin microcirculatory perfusion indices and biomarkers of endothelial dysfunction in patients with septic shock: an observational study. Crit Care 23: 311. [View]
- Varga Z, Flammer AJ, Steiger P, et al. (2020) Endothelial cell infection and endotheliitis in COVID-19. The Lancet 395: P1417–1418. [View]
- Wiersinga WJ, Rhodes A, Cheng AC, et al. (2020) Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19). JAMA 324: 782–793. [View]
- Ackermann M, Verleden SE, Kuehnel M, et al. (2020) Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N Engl J Med 983: 120–128. [View]
- Poissy J, Goutay J, Caplan M, et al. (2020) Pulmonary embolism in patients with COVID-19: Awareness of an increased prevalence. Circulation 142: 184–186. [View]
- Sinha P, Matthay MA, Calfee CS (2020) Is a “cytokine storm” relevant to COVID-19. JAMA Int Med 180: 1152–1154. [View]
- Tang N, Bai H, Chen X, et al. (2020) Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost 18: 1094–1099. [View]
- Wu Y, Wang T, Guo C, et al. (2020) Plasminogen improves lung lesions in patients with COVID-19. QJM 113: 539–545. [View]
- Moore HB, Barrett CD, Moore EE, et al. (2020) Is there a role for tissue plasminogen activator as a novel treatment for refractory COVID-19 associated acute respiratory distress syndrome? J Trauma Acute Care Surg 88: 1–2. [View]
Supplementary Material:
Figure 2 and figure 3 demonstrate the exaggerated and prolonged inflammatory reaction in patients with invasive mechanical ventilation in comparison to patients in the NIV-G. In patients with invasive mechanical ventilation, CRP and IL-6 were still elevated after 28 days demonstrating that the SARS-CoV-2 virus can induce a massive and prolonged systemic inflammatory response in some patients.
Median values and 25th and 75th quartiles are shown for the IV-G and the NIV-G
Note: Upper limit for CRP in our laboratory is < 5mg/l ; Upper limit for Interleucin-6 in our laboratory is < 7pg/ml
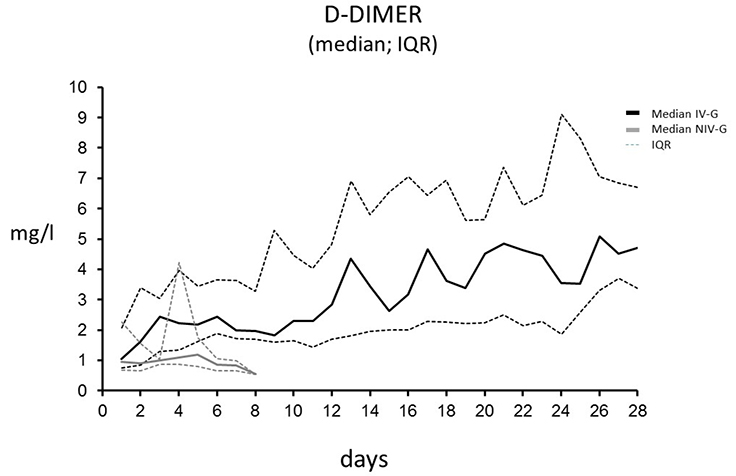
Figure 4 shows D-Dimer values in patients with invasive mechanical ventilation in comparison to patients in the NIV-G. In contrast to patients in the NIV-G, patients with invasive mechanical ventilation demonstrate prolonged coagulation activation and fibrinolysis. In most patients in the IV-G, D-Dimer serum concentrations were still rising after 28 days.
Median values and 25th and 75th quartiles are shown for the IV-G and the NIV-G
Note: Upper limit for D-Dimer in our laboratory is < 0,5mg/l
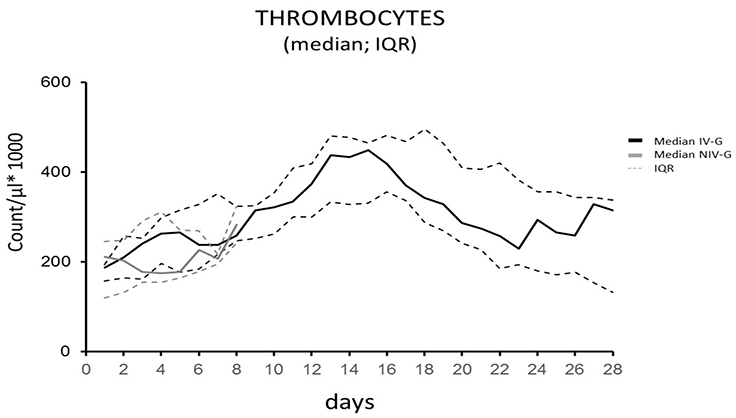
Figure 5 illustrates thrombocyte count in IV-G and NIV-G patients. Despite massive activation of fibrinolysis, mild thrombocytopenia with abnormal thrombocyte count occurred only in a minority of patients.
Median values and 25th and 75th quartiles are shown for the IV-G and the NIV-G
Note: Normal thrombocyte count in our laboratory is 182-369 (x1000)/µl
Article Type
Research Article
Publication history
Received: October 21, 2021
Accepted: November 11, 2021
Published: November 17, 2021
Citation:
Hasibeder W, Perschinka F, Krismer C, Wöll E (2021) Severe Covid-19 Disease – An Unusual Type of Skin Mottling. Clar J Infect Dis Ther 02(01): 91–98.
Walter Hasibeder1*, Fabian Perschinka1, Christoph Krismer2, and Ewald Wöll2
1Department of Anesthesiology and Intensive Care Medicine, St. Vinzenz Krankenhaus, Sanatoriumsstraße 43, 6511 Zams, Austria
2Department of Internal Medicine, St. Vinzenz Krankenhaus, Sanatoriumsstraße 43, 6511 Zams, Austria
*Corresponding author
Walter Hasibeder, MD,
Dept. of Anesthesiology and Intensive Care Medicine,
St. Vinzenz Krankenhaus
Sanatoriumsstraße 43,
6511 Zams,
Austria;