- 4192 Devonwood Way, Ashburn, Virginia, 20148
- Helpline: +1 (703) 665-3747
Summary and Case Observation of Tumour Treating Fields Therapy for Glioblastoma
- Home
- Back to Journal
- Article Details
Introduction
Glioblastoma (GBM) is s a highly invasive common tumor of the central nervous system account for about 50% of the primary malignant tumors in the brain, with a high degree of malignancy and a poor prognosis [1]. Although At present, the standard treatment including is maximum resection, combined with radiotherapy and temozolomide chemotherapy are performed, but the median survival time of GBM patients is still only 14 to17 months, and the 5-year survival rate is 9.8% [2]. How to improve the effectiveness of glioblastoma treatment, scientists and clinicians have made many attempts, but the results are not good [3-8]. No other new drug therapies have been available [9] ever since temozolomide was marketed in 2005, and the US Food and Drug Administration (FDA) approved bevacizumab for patients with recurrent glioblastoma. The drug treatment of glioblastoma has encountered difficulties; however the scientists have discovered a new treatment method, which is Tumour Treating Fields (TTF).
In 2004, the first paper about TTF was published. Low-intensity, intermediate-frequency (100-300 kHz), alternating electric fields, delivered by means of insulated electrodes, were found to have a profound inhibitory effect on the growth rate of a variety of human and rodent tumor cell lines and malignant tumors in animals [10]. In 2008, studies have reported that 6 cases of breast cancer skin metastasis and melanoma skin metastasis patients underwent TTF, the progress of the lesions was significantly controlled [11]. In 2009, the application of TTF in the treatment of recurrent glioblastoma multiforme was reported, the overall survival of patients treated with TTF reached 62 weeks [12]. Based on the excellent experimental research results, the FDA approved TTF as a treatment plan for recurrent gliomas in 2011. The first paper on Phase III clinical trial was published in 2012. The study, which included 237 patients with recurrent glioblastoma, showed that TTF alone could achieve the same treatment effect as chemotherapy, and the patients’ quality of life was better than that of the chemotherapy group [13]. In 2013, TTF was written into the Clinical Practice Guidelines for Cancer Diagnosis and Treatment by the National Comprehensive Cancer Network (NCCN) [14]. In 2014, Stupp published study on a prospective phase III randomized controlled trial again. It collected 315 patients with newly diagnosed glioblastoma, the Progression-Free Survival Time (PFS) and Overall Survival time (OS) of patients treated with temozolomide combined with TTF were about 3 months longer than those treated with temozolomide alone [15]. In October 2015, TTF was further approved by the FDA for the treatment of newly diagnosed glioblastoma. In 2017, the latest blockbuster data on TTF clinical trials were released, with 695 participants divided into two groups, one using temozolomide alone and another group using temozolomide combined with TTF. The results showed that the patients’OS with TTF combined with chemotherapy was 5 months longer than that of chemotherapy alone [15]. In 2018, the NCCN guidelines listed TTF as a category 1 recommendation [16]. In December 2018, the guidelines for diagnosis and treatment of brain Glioma (2018 Edition) issued by the National Health Commission of China also specially recommended TTF for newly diagnosed glioblastoma and recurrent high-grade glioma [17].
At the end of 2018, TTF entered Hong Kong, and Chinese patients began to have the opportunity to accept this advanced treatment. There are not many users in the inland areas of mainland China because of various factors. Fortunately, we collected a patients with glioblastoma are currently receiving TTF treatment in West China Hospital of Sichuan University. She went to Hong Kong to start using TTF on July 4, 2019. After about 6 months of follow-up, the following is a preliminary report on the short-term efficacy and adverse reactions of this case.
Case
A 54-year-old female, developed a headache in December 2017. Examination showed that the left parietal occipital lobe occupied, and the first surgical treatment was performed, and the postoperative pathological diagnosis was glioblastoma. After operation, the patient received routine radiotherapy, 60Gy/33f, and temozolomide chemotherapy, 100mg/d (during radiotherapy), 300mg d1-5, q4w. In January 2019, the patient reexamined the MRI and found that the left temporal parietal space was occupied (Figure 1) and was operated again. The postoperative pathological diagnosis was still glioblastoma. The patient had occasional headache after operation, and the head MRI examination three months after operation indicated the progress of the disease (Figure 2). TTF was prepared to be used, and finally officially used the TTF treatment on July 4, 2019. The use time was relatively fixed, the utilization rate was 93%, and the compliance was good (Figure 3). During treatment, the patient developed allergic rash scattered on the scalp, mild itching, no ulceration and infection, and recovered quickly after topical application of antiallergic drugs. There had been several times that the patch temperature was too high, and the reasons are as follows: 1. The ambient temperature used was high (e. G. hot weather, direct sunlight, etc.). 2. The contact between the patch and the skin was not good. By instructing the patient to adjust the indoor temperature to about 24 °C, to use the headband or tape to strengthen the loose patch, to use the headband when sleeping at night, and to pay attention to clean the hair of the scalp every time, the situations had been significantly improved. Re-examination of the MRI in October 2019 showed that the condition was stable (Figure 4). Follow-up to 2020.1.23, the patient showed no symptoms of discomfort.
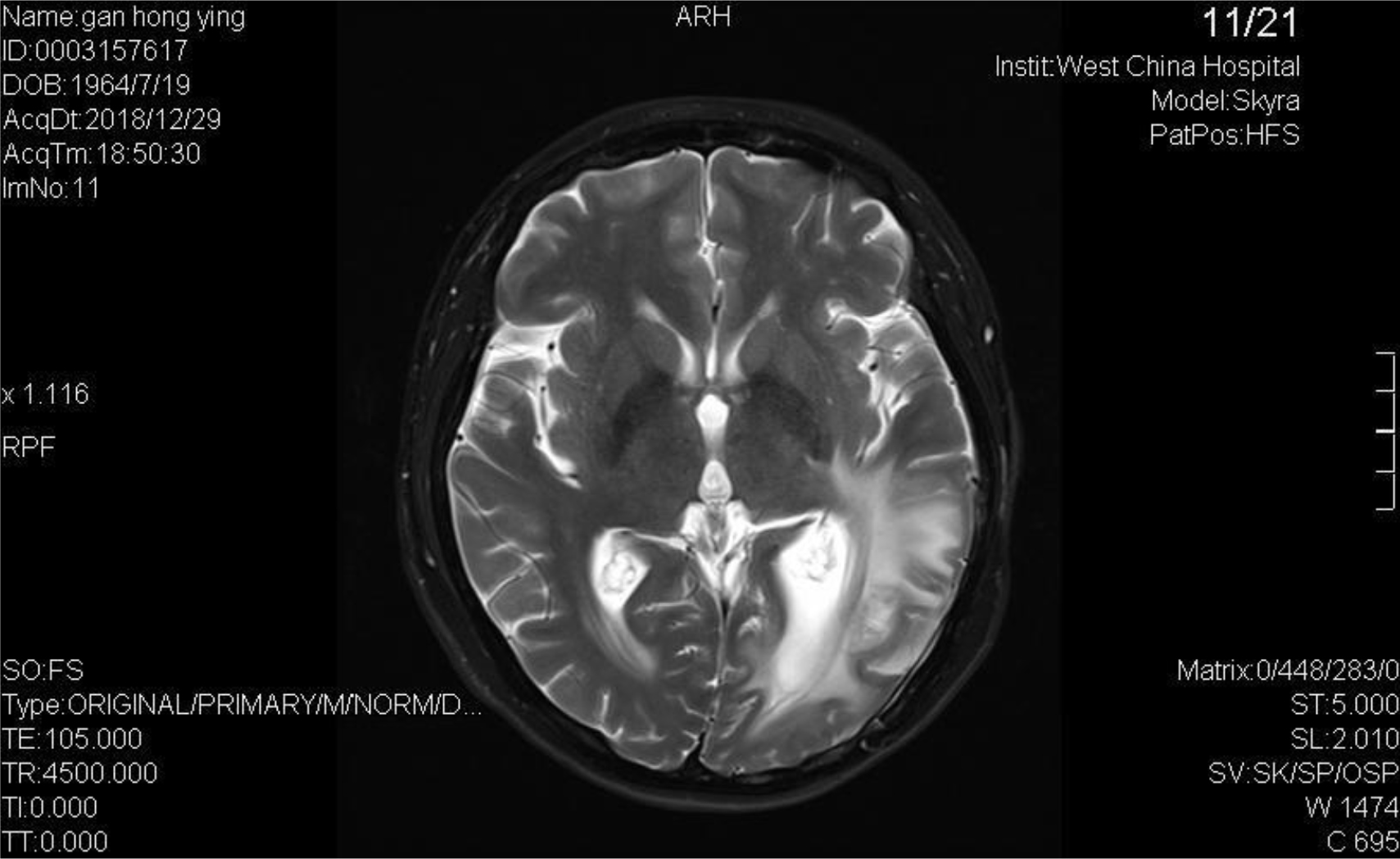
Figure 1
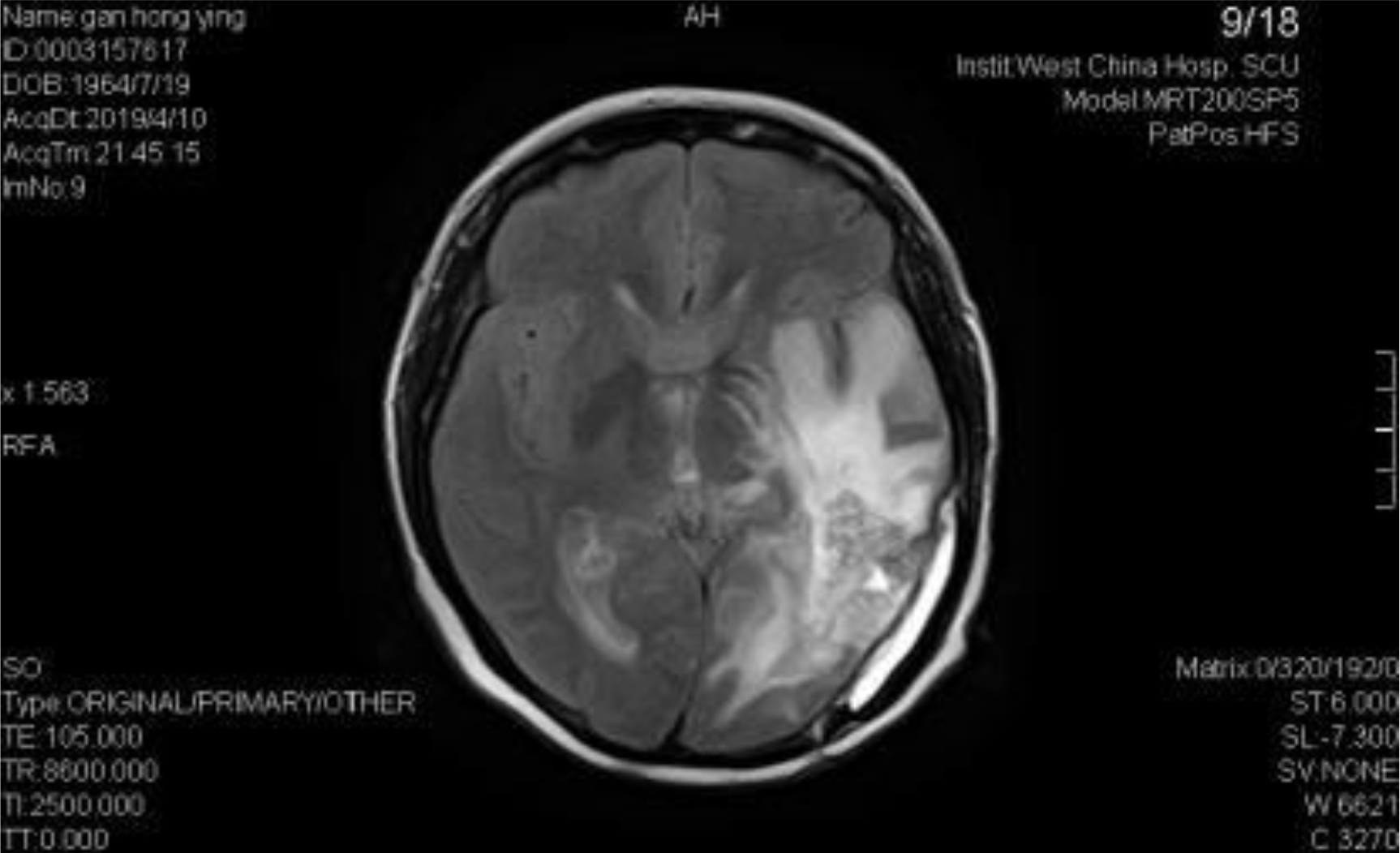
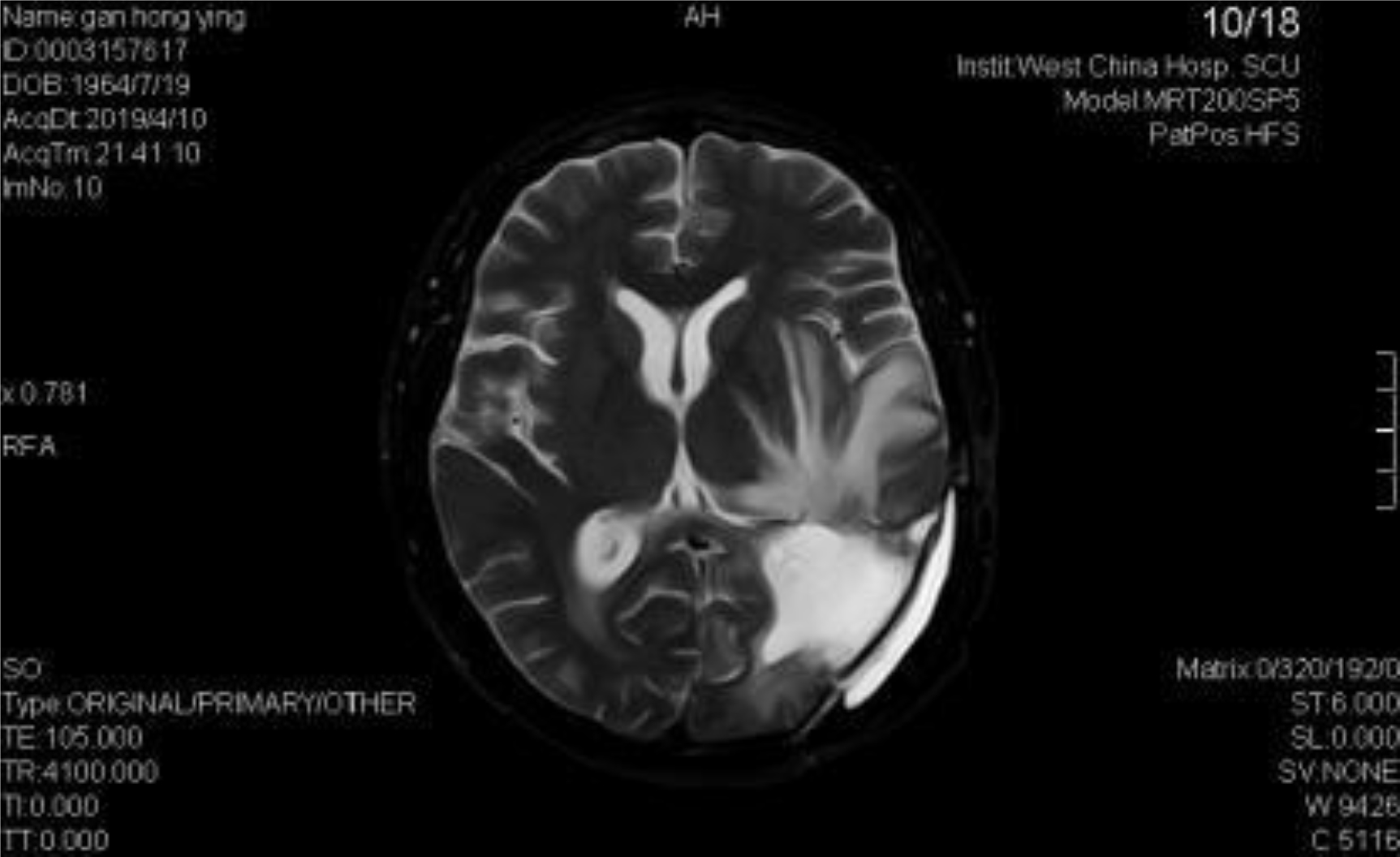
Figure 2
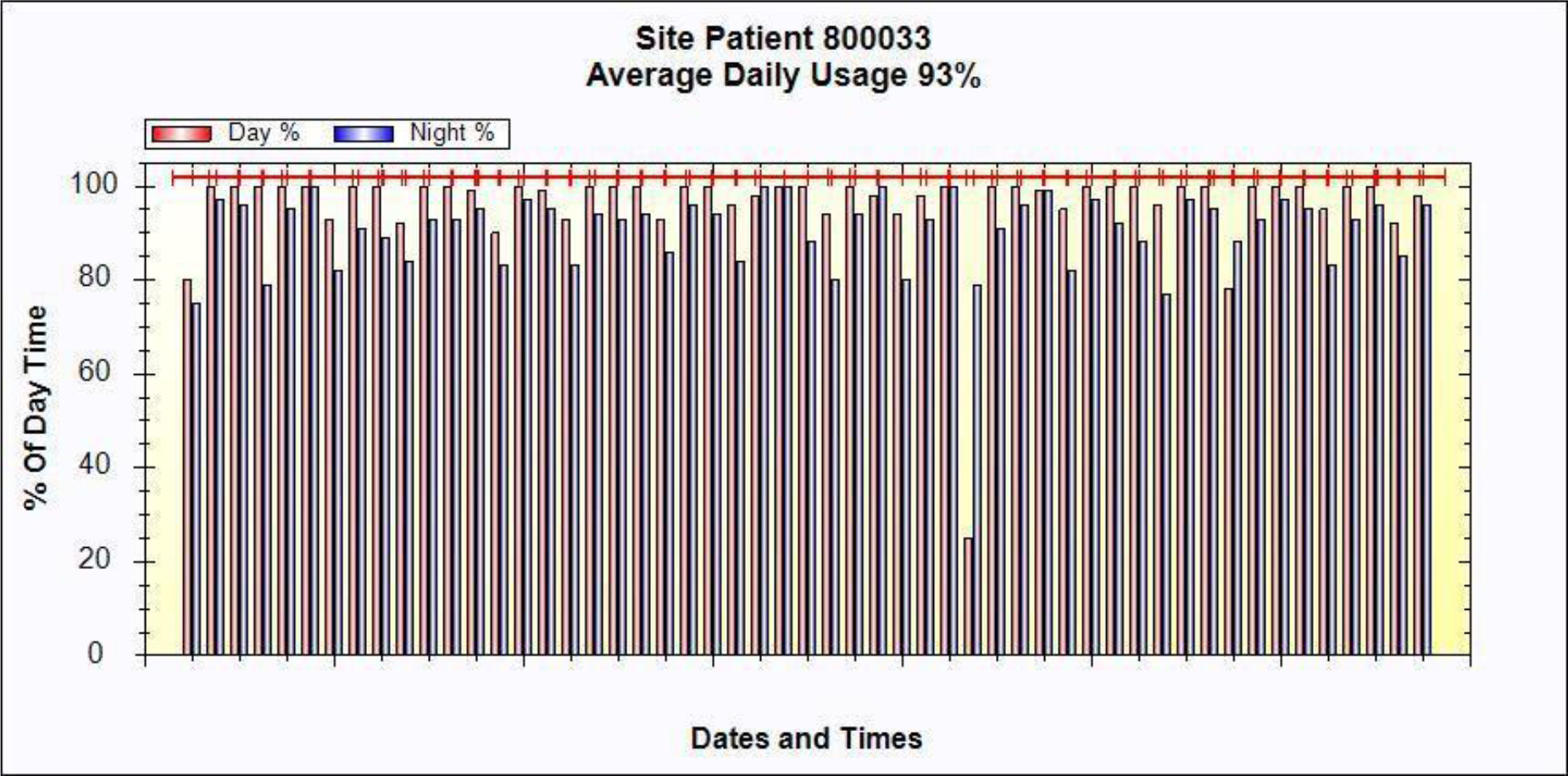
Figure 3
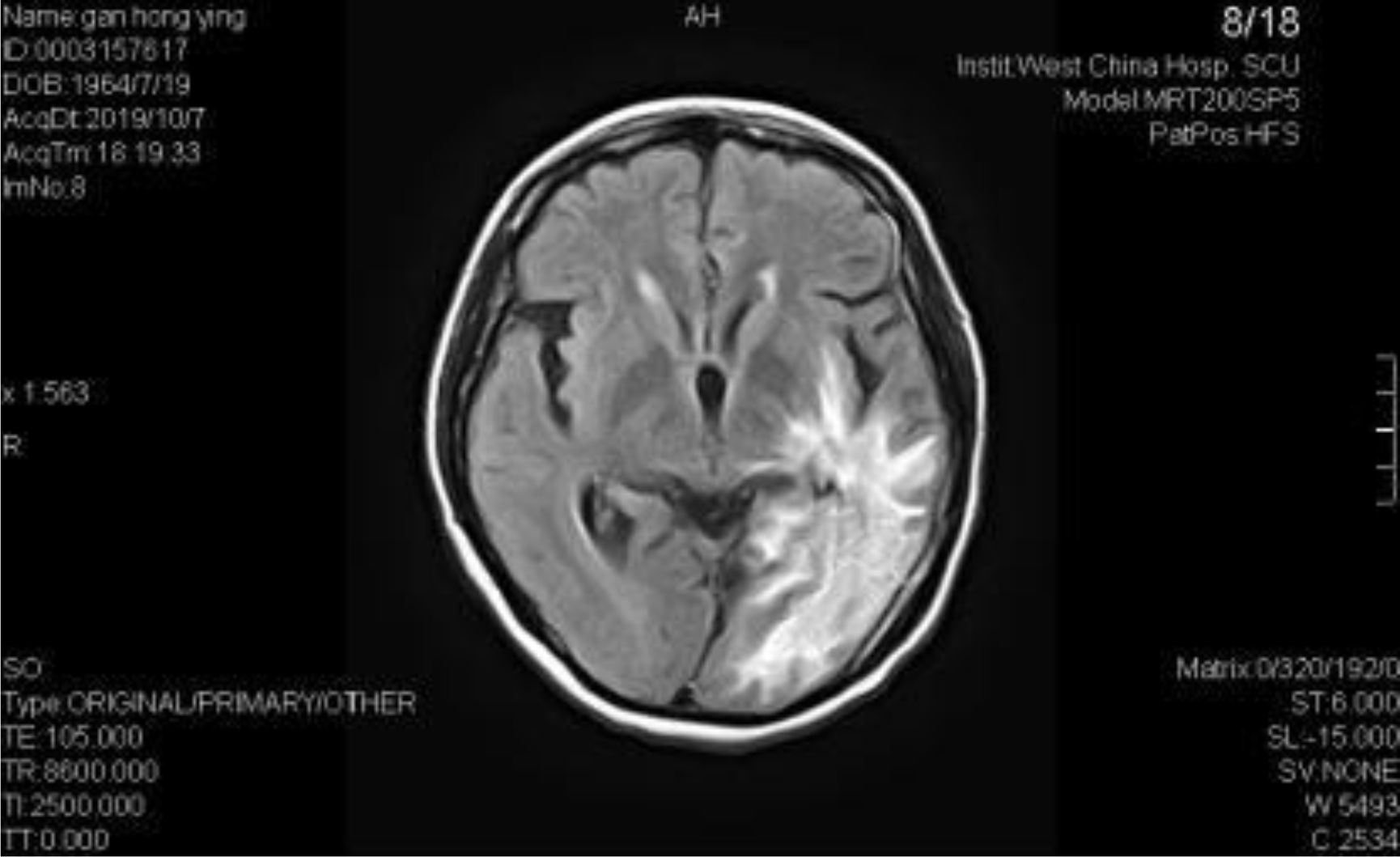
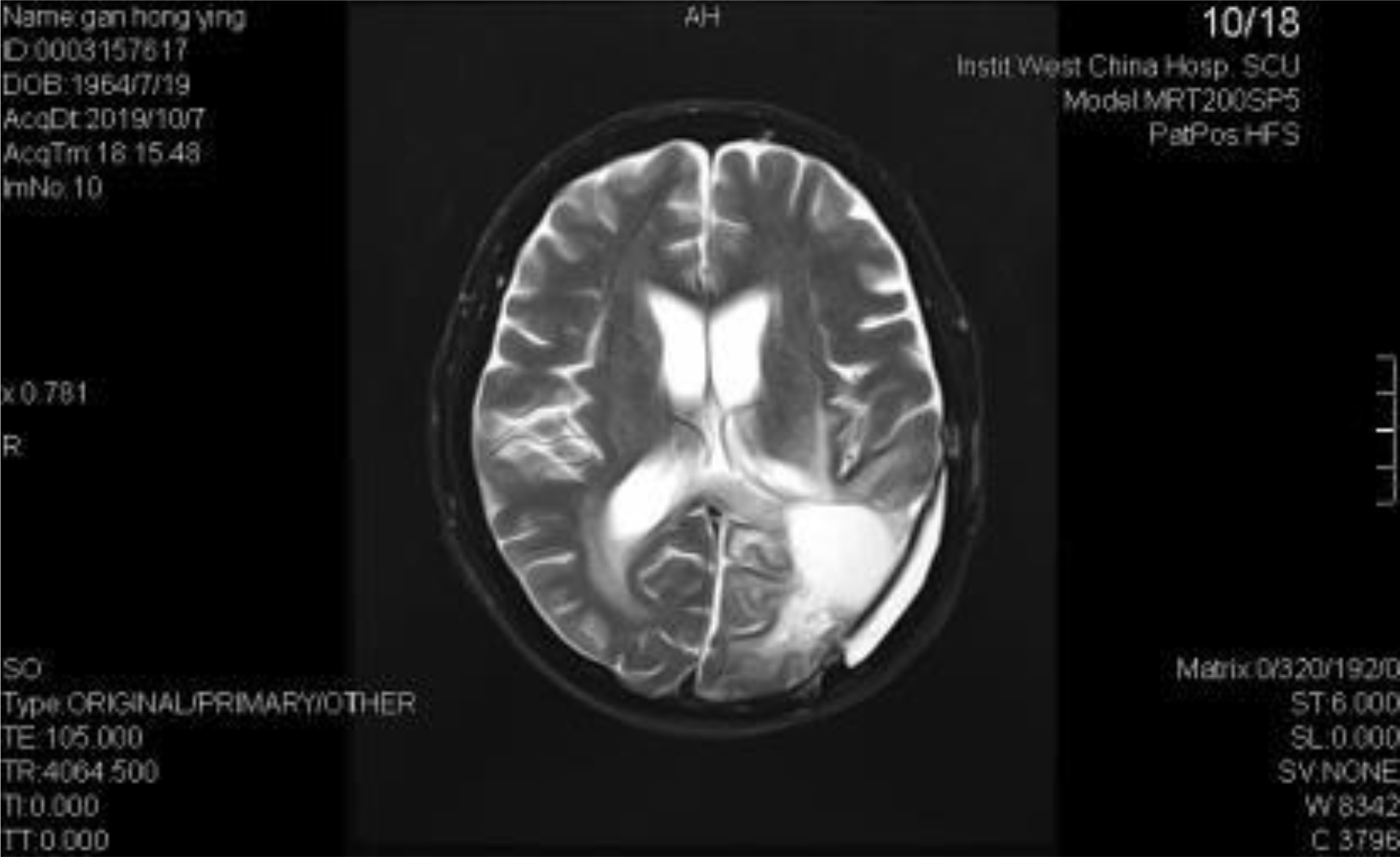
Figure 4
TTF has no side effects such as alopecia, vomiting, fatigue, anemia, cognitive function or neurological impairment caused by traditional radiotherapy and chemotherapy. It can not only improve the PFS, but also prolong the OS. TTF has been demonstrated to have excellent effectiveness and tolerance. As a new anti-tumor therapy, TTF has not been developed for a long time, and further research is needed to improve the clinical application. At present, many studies are actively looking for the optimal intensity and frequency of treatment, as well as the use model combined with immune checkpoint inhibitors or targeted drugs, and determines the best efficacy and safety of this combination model. These opinions still need to be studied in clinical trials with large number of samples. At present, TTF has come to mainland China. It is expected that with the efforts of all sides, TTF will be as feasible as traditional radiotherapy and chemotherapy to benefit more Chinese patients.
Acknowledgement
Research and development of tumor real-time monitoring molecular diagnostic products based on liquid biopsy -a major science and technology project of Guangdong province 2019B020232003.
Dalian municipal Science and technology innovation projects (2018 j12sn063): a new method for the detection optical flow control chip peripheral blood tumor cells research Science and technology innovation project of Dalian City (No: 2018 j12sn063).
This project is funded by the research and data on the evaluation method of stereotactic radiotherapy equipment (subject no: 2017YFC0113701).
References
- Rl S, Kd M, AJ (2019) Cancer statistics, 2019. CA: A cancer journal for clinicians 69: 7–34. [view]
- Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, et al. (2009) Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. The Lancet Oncology 10: 459–466. [view]
- Wen PY, Touat M, Alexander BM, Mellinghoff IK, Ramkissoon S, et al. (2019) Buparlisib in Patients With Recurrent Glioblastoma Harboring Phosphatidylinositol 3-Kinase Pathway Activation: An Open-Label, Multicenter, Multi-Arm, Phase II Trial. J Clin Oncol 37: 741–750. [view]
- Wick W, Dettmer S, Berberich A, Kessler T, Karapanagiotou-Schenkel I, et al. (2019) N2M2 (NOA-20) phase I/II trial of molecularly matched targeted therapies plus radiotherapy in patients with newly diagnosed non-MGMT hypermethylated glioblastoma. Neuro Oncol 21: 95–105. [view]
- Taylor JW, Parikh M, Phillips JJ, James CD, Molinaro AM, et al. (2018) Phase-2 trial of palbociclib in adult patients with recurrent RB1-positive glioblastoma. J Neurooncol 140: 477–483. [view]
- Weller M, Butowski N, Tran DD, Recht LD, Lim M, et al. (2017) Rindopepimut with temozolomide for patients with newly diagnosed, EGFRvIII-expressing glioblastoma (ACT IV): a randomised, double-blind, international phase 3 trial[J]. Lancet Oncol 18: 1373–1385. [view]
- Di Stefano AL, Fucci A, Frattini V, Labussiere M, Mokhtari K, et al. (2015) Detection, Characterization, and Inhibition of FGFR-TACC Fusions in IDH Wild-type Glioma. Clin Cancer Res 21: 3307–3317. [view]
- D S, M M, A K, et al. (2020) Insights in the immunobiology of glioblastoma. Journal of molecular medicine (Berlin, Germany), 98: 1–10.
- Patel MAE (2014) Cancers. 6: 1953–1985.
- Ed K, Z G, R S, et al. (2004) Disruption of cancer cell replication by alternating electric fields. Cancer research 64: 3288–3295.
- Salzberg M, Kirson E, Palti Y, Rochlitz C (2008) A pilot study with very low-intensity, intermediate-frequency electric fields in patients with locally advanced and/or metastatic solid tumors. Onkologie 31: 362–365. [view]
- Kirson ED1, Schneiderman RS, Dbalý V, Tovarys F, Vymazal J, et al. (2009) Chemotherapeutic treatment efficacy and sensitivity are increased by adjuvant alternating electric fields (TTFields). BMC medical physics 9: 1. [view]
- Stupp R, Wong ET, Kanner AA, Steinberg D, Engelhard H, et al. (2012) NovoTTF-100A versus physician’s choice chemotherapy in recurrent glioblastoma: a randomised phase III trial of a novel treatment modality. Eur J Cancer 48: 2192–2202. [view]
- Network. N C C. Central Nervous System Cancers (Version 1.2013). [view]
- Stupp R W E, Scott C, et al. (2014) Interim Analysis of the EF-14 Trial: A Prospective, Multi-center Trial of NovoTTF-100A Together With Temozolomide Compared to Temozolomide Alone in Patients with Newly Diagnosed GBM. Neuro-oncology 16: 167.
- Network. N C C. Central Nervous System Cancers (Version 1.2018).
- China NHCO (2018) the guidelines for diagnosis and treatment of brain Glioma (2018 Edition).
Article Type
Case Report
Publication history
Received: March 07, 2023
Accepted: March 12, 2023
Published: March 14, 2023
Citation:
L Zeng, X-Q Xie, D-B Wu, Y-Z Liu, J-B Kang, et al. (2023) Summary and Case Observation of Tumour Treating Fields Therapy for Glioblastoma. Cancer Stud and Therap I, Volume 01(01): 1–5.
L Zeng1, X-Q Xie2, D-B Wu3, Y-Z Liu4, J-B Kang5, B-L Qu6, P Ai1, Q Mao9, Y-H Liu9, X Wang9, Y Luo1, L Xie1, P Li1, L Bai1, F Wang1*, X.-F Li7*, J-J Wang8* and W-M Li10
1Department of Medical Oncology, Cancer Center, West China Hospital, West China Medical School, Sichuan University, Sichuan, P.R. China
2Department of Critical Care Medicine, West China Hospital, Sichuan University, Sichuan, P.R. China
3Cancer Hospital, Ansteel Group Hospital, Anshan, Liao Ning, P.R. China
4Department of Radiotherapy, Beijing Hospital, Beijing, P.R. China
5Department of Radiotherapy, The Sixth Medical Center of PLA General Hospital, Beijing, P.R. China
6Department of Radiotherapy, Chinese PLA General Hospital, Beijing, P.R. China
7Department of Radiotherapy, First Hospital of Shanxi Medical University, Taiyuan, Shanxi, P.R. China
8Department of Radiation Oncology Cancer Center, Peking University 3rd Hospital, P.R. China
9Department of Neurosurgery, West China Hospital, Sichuan University, Chengdu, China
10Precision medicine center, West China Hospital, West China Medical School, Sichuan University, Sichuan, P.R. China
*Corresponding authors
Feng Wang,
Department of Medical Oncology,
West China Hospital,
West China Medical School,
Sichuan University, No. 37,
Guo Xue Xiang Street.
Chengdu 610041,
Sichuan Province, China;
Phone: 008618980602023;
Fax: 0086-28-85423278;
Xian Feng Li,
Department of Radiotherapy,
First Hospital of Shanxi Medical University,
Taiyuan, Shanxi,
P.R. China;
Jun Jie Wang,
Department of Radiation Oncology Cancer Center,
Peking University 3rd Hospital,
P.R. China;